Repeat the calculations of Example 12.1 but use 1 = methanol, 2 = water, and 3 =
Question:
Repeat the calculations of Example 12.1 but use 1 = methanol, 2 = water, and 3 = acetone. Are the results any different? If not, why not? Prove your conclusion mathematically.
EXAMPLE 12.1
This example is similar to Example 11.5.1 on page 283 of Taylor and Krishna [15]. The following results were obtained for Tray n from a rate-based calculation of a ternary distillation at 14.7 psia, involving acetone
(1), Methanol
(2), And water
(3) In a 5.5-ft-diameter column using sieve trays with a 2-inch-high weir. Vapor and liquid phases are assumed to be completely mixed.
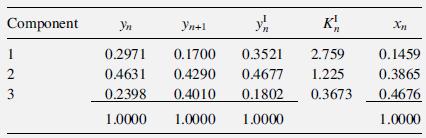
The computed products of the gas-phase, binary mass-transfer coefficients and interfacial area, using the Chan–Fair correlation of §6.6, are as follows in lbmol/(h-unit mole fraction):
k12 = k21 = 1; 955; k13 = k31 = 2; 407; k23 = k32 = 2; 797
(a) Compute the molar diffusion rates.
(b) Compute the masstransfer rates.
(c) Calculate the Murphree vapor-tray efficiencies.
Step by Step Answer:

Separation Process Principles Chemical And Biochemical Principles
ISBN: 9780470481837
3rd Edition
Authors: By J. D. Seader, Ernest J. Henley, D. Keith Roper





