Name each of the compounds below, using the IUPAC system of nomenclature. CI OH () (b) (d)
Question:
Name each of the compounds below, using the IUPAC system of nomenclature.
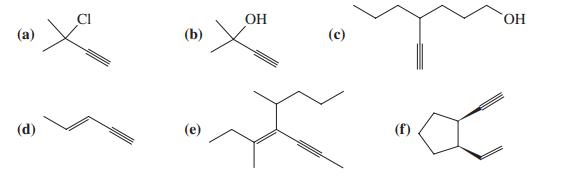
Transcribed Image Text:
CI OH (а) (b) (d) (е) (f)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
Q Name each of the compounds fellow using the IUPAC sy...View the full answer

Answered By

Seenaiah Gangipaka
I am post graguate(MSc-Organic Chemistry) and B.Ed(Physical Science).I have 11 years of good experience in teaching and professional.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Name each of the compounds below. Use cis / trans and/or E/Z designations, if appropriate, to designate stereochemistry a. b. c. Cl C=C H,C CH3 CH2CI H3C H3C H,C Cl
-
Name each of the following compounds according to substitutive IUPAC nomenclature: (a) (CH3)2CHCH2CH2CH2Br (b) (CH3)2CHCH2CH2CH2OH (c) Cl3CCH2Br (d) (e) CF3CH2OH (f) (g) (h) (i) Cl2CHCHBr CI OH ,
-
For each species in Problem 32, indicate the resonance form that is the major contributor to the resonance hybrid. Explain your choices. Data From Problem 32 CH3 () (b) () (d)
-
What were the reasons for the failure of the original purpose of FordDirect.com? Who eventually gained from the system and what were the gains?
-
Explain the potential impact to project costs and schedules when assigning the following task relationships (mandatory, discretionary, and external).
-
Modify the program you wrote for Programming Challenge 2 (Distance Traveled) so it writes the report to a file instead of the screen. Open the file in Notepad or another text editor to confirm the...
-
Profit sharing has been promoted by governments for many years. The Share Incentive Plan is the latest attempt to encourage employees to hold shares in their own companies.LO1
-
Robertos Honey Farm in Chile makes five types of honey: cream, filtered, pasteurized, mlange (a mixture of several types), and strained, which are sold in 1 or 0.5 kg glass containers, 1 kg and 0.75...
-
(The Binomial Model) A non-dividend paying stock is trading at $50. The continuously compounded interest rate is 2% for all maturities. Assume the stocks annualized volatility is 30%. Use a 2-period...
-
(a) Use the northwest-corner method to find an initial feasible solution to the following problem. What must you do before beginning the solution steps? (b) Use the intuitive lowest-cost approach to...
-
Draw the structures of the molecules with the following names. (a) 1-Chloro-1-butyne (b) (Z)-4-Bromo-3-methyl-3-penten-1-yne (c) 4-Hexyn-1-ol
-
Compare C H bond strengths in ethane, ethene, and ethyne. Reconcile these data with hybridization, bond polarity, and acidity of the hydrogen.
-
An experiment was conducted using a randomized block design. The data from the experiment are displayed in the following table. a. Fill In the missing entries in the ANOVA table. b. Specify the null...
-
1. What is DBMS and what is its utility? Explain RDBMS with examples. 2. What is a Database? 3. Mention the issues with traditional file-based systems that make DBMS a better choice 4. Explain a few...
-
1. State the difference between lists and tuples in Python programming. 2. Explain why Python is an Interpreted Language
-
1. How does Python handle memory? 2. Python's ternary operators: how do they work? 3. How is Python's multithreading implemented
-
In a relational database, explain the difference between Inner join & Outer join. Provide an example query for each and describe the result set produced by each query.
-
Consider a class Characteristic that will be used in an online dating service to assess how compatible two people are. Its attributes are descriptiona string that identifies the characteristic...
-
Following is the current balance sheet for a local partnership of doctors: The following questions represent independent situations: a. E is going to invest enough money in this partnership to...
-
We remarked in Impact 19.2 that the particle in a sphere is a reasonable starting point for the discussion of the electronic properties of spherical metal Nan particles. Here, we justify eqn 9.54,...
-
Outline the electron configurations of many-electron atoms in terms of their location in the periodic table.
-
Describe the separation of variables procedure as it is applied to simplify the description of a hydrogenic atom free to move through space.
-
I am struggling with this and need help desperately thank you! June 1: Byte of Accounting, Inc, issued 2,025 shares of its common stock after acquiring from Courtney $42,050 in cash, computer...
-
Watercraft s predetermined overhead rate is 2 0 0 % of direct labor. Information on the company s production activities during May follows. Purchased raw materials on credit, $ 2 4 0 , 0 0 0 ....
-
Required information Skip to question [ The following information applies to the questions displayed below. ] Iguana, Inc., manufactures bamboo picture frames that sell for $ 2 0 each. Each frame...

Study smarter with the SolutionInn App


