For each species in Problem 32, indicate the resonance form that is the major contributor to the
Question:
For each species in Problem 32, indicate the resonance form that is the major contributor to the resonance hybrid. Explain your choices.
Data From Problem 32
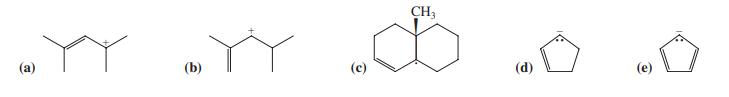
Transcribed Image Text:
CH3 (а) (b) (с) (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
The option e is more stable resonating structuret...View the full answer

Answered By

Manoj Upadhyay
I have teaching experience of 11 years.I have all abilities to solve the problem of chemistry.I always practice of short cuts for various problems.I also work as SME in various institution.I have worked as lecturer in many reputed college. I also worked as IIT OrJEE expert teacher.I believe that education is the best tool in order to get success.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
For matrix P form problem 32 with (A) S0 =[0 0 1] (B) S0 - [.2 .5 .3] Problems 35-40 refer to the matrices in Problems 29-34. Use the limiting matrix P found for each transition matrix P in Problems...
-
For each set of resonance structures that follow, add a curved arrow that shows how electrons in the left formula shift to become the right formula, and designate the formula that would contribute...
-
From each set of resonance structures that follow, designate the one that would contribute most to the hybrid and explain your choice: (a) (b) (c) (d) (e) (f) HO HO
-
Write a text-based transaction program for recording stock levels in a warehouse. A transaction is either an import, an export, or wastage (e.g., when stock falls off a shelf and breaks) of items,...
-
List and briefly explain the three key deliverables for the scope planning process.
-
In January 2022, Mr. D, who is self-employed, purchased a new automobile, which he uses 100% for business. During 2022, he drove the car 14,000 miles. Half of those miles were driven January through...
-
An income statement for Cheries Pizza is included in the Working Papers. 1. Use the following information to calculate the breakeven point in sales dollars and unit sales for July. The August...
-
How much acquisition and integration cost was included within GAAP Other (expense) income, net for the 6 months ended December 31, 2018? $0 $1,755 $2,613 $4,368
-
how you would use this information to create your business plan or use this information to assist with your business needs with Chp 5 in MGMT 2145 Business Plan Development. In Book(BARINGER,...
-
A carbon/epoxy lamina is clamped between rigid plates in a mold (see figure below) while curing at a temperature of 125C. After curing, the lamina/mold assembly (still clamped together) is cooled...
-
Name each of the compounds below, using the IUPAC system of nomenclature. 28. Name each of the compounds below, using the IUPAC system of nomenclature. CI OH (a) (b) HO. (c) (d) 3 (e) (f)
-
Illustrate by means of appropriate structures (including all relevant resonance forms) the initial species formed by (a) breaking the weakest C H bond in 1-butene; (b) treating 4-methylcyclohexene...
-
Go to http://finance.yahoo.com/, enter the symbol HFWA (Heritage Financial Corporation), and click on Get Quotes. Then, retrieve stock price data at the beginning of the last 20 quarters. Then go to...
-
Using the EEO-1 report as an example, discuss the purpose of government mandates. Give examples of penalties that organizations incur when they fail to comply with government mandates such as EEO and...
-
What is generalised audit software? Identify the four main substantive procedure applications using this software.
-
. What are the three main CAATs used in testing controls? Compare and contrast these techniques.
-
What specific knowledge about computer controls should the auditor acquire in obtaining an understanding?
-
What are some of the differences between using computers and using a manual system on an entitys internal controls?
-
Determine the carburizing time necessary to achieve a carbon concentration of 0.30 wt% at a position 4 mm into an iron-carbon alloy that initially contains 0.10 wt% C. The surface concentration is to...
-
The executor of Gina Purcells estate has recorded the following information: Assets discovered at death (at fair value): Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Calculate the relative numbers of Br, molecules (v = 321 cm-1) in the second and first excited vibrational states at (a) 298 K, (b) 800 K.
-
Prom the data in Exercise 13.18a, predict the fundamental vibrational Wave numbers of the deuterium halides.
-
For 14N2 G values for the transitions v = 1 0, 2 0, and 3 0 are, respectively, 2345.15, 4661.40, and 6983.73 cm-1. Calculate v and xe- Assume ye to be zero.
-
Cori's Dog House is considering the installation of a new computerized pressure cooker for hot dogs. The cooker will increase sales by $7,900 per year and will cut annual operating costs by $14,000....
-
Riley Co experiences 20 million loss this year. It is more than likely than not that this Net operating loss will expire unused. How should Riley record this loss as part of calculating its income...
-
( Fixed Income Securities ) 1 8 ) F ) Define the curvature of the term structure with respect to short - term, medium - term and long - term bonds and hence calculate the curvatures of the term...

Study smarter with the SolutionInn App


