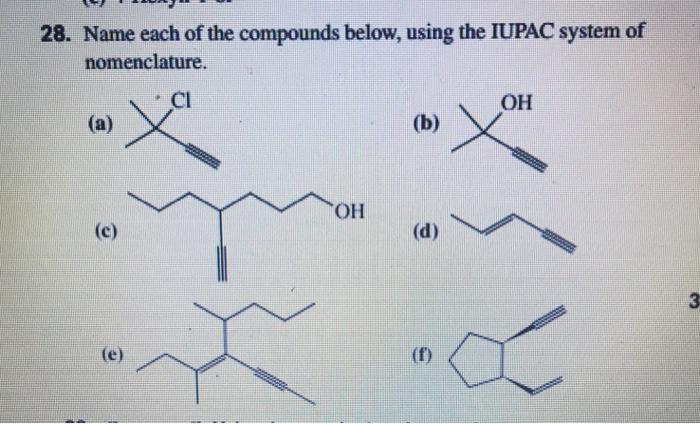
Name each of the compounds below, using the IUPAC system of nomenclature. 28. Name each of the
Question:
Name each of the compounds below, using the IUPAC system of nomenclature.
Transcribed Image Text:
28. Name each of the compounds below, using the IUPAC system of nomenclature. CI OH (a) (b) HO. (c) (d) 3 (e) (f)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
a 3 chloro 3 methyl but 1yne b 11 ...View the full answer

Answered By

YOGESH JANGID
I am NIT SRINAGAR student please check my all NRAF ranking of my college . I am chemical engineering student and I good scored in 1st year and I good helpful to you all. I scored good in IIT college .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Name each of the compounds below. Use cis / trans and/or E/Z designations, if appropriate, to designate stereochemistry a. b. c. Cl C=C H,C CH3 CH2CI H3C H3C H,C Cl
-
Name each of the following compounds according to substitutive IUPAC nomenclature: (a) (CH3)2CHCH2CH2CH2Br (b) (CH3)2CHCH2CH2CH2OH (c) Cl3CCH2Br (d) (e) CF3CH2OH (f) (g) (h) (i) Cl2CHCHBr CI OH ,
-
For each species in Problem 32, indicate the resonance form that is the major contributor to the resonance hybrid. Explain your choices. Data From Problem 32 CH3 () (b) () (d)
-
On January 1, 2014, Ultra Green Packaging purchased a used machine for $156,000. The next day, it was repaired at a cost of $4,068 and mounted on a new platform that cost $5,760. Management estimated...
-
List and explain the characteristics that determine the size and depth of the scope statement.
-
Salomon has been approved for a car loan from Royal Bank $ 2 1 1 3 7 at 6 . 5 % compounded monthly. What end - of - month payments are required to pay off the loan in 4 years? What is N , I / Y , C /...
-
Use the working papers and data from Work Together 15-2. Forms for completing this Work Together are provided in the Working Papers. Your instructor will guide you through the following examples. 1....
-
1. Povero believes that interest rates are a major factor in the real estate market. What are the implications of rising, falling, and steady interest rates for future real estate prices? 2. If the...
-
Detailed marketing strategy in line with market penetration strategy for a food and drinks company opening new stores in underserved areas.
-
1. Brooking agreed to sell a tract of land to Dover Pool & Racquet Club, Inc., on which Dover planned to build a swim and tennis club. Neither party to the contract knew that just before the contract...
-
Draw all resonance forms and a representation of the appropriate resonance hybrid for each of the following species. CH3 () (b) () (d) ()
-
Illustrate by means of appropriate structures (including all relevant resonance forms) the initial species formed by (a) breaking the weakest C H bond in 1-butene; (b) treating 4-methylcyclohexene...
-
Compare the temperatures at which soda-lime, borosilicate, 96% silica, and fused silica may be annealed.
-
Define the term tolerable deviation rate and discuss the factors that should be considered in deciding on the tolerable rate for both substantive procedures and tests of controls.
-
What are the basic similarities and differences between statistical and non-statistical sampling?
-
Discuss the main risks associated with EDI systems.
-
Identify the key words and phrases in the auditors report that describe the character of the audit.
-
What is microcomputer-based audit software? Identify the common applications of this software.
-
An FCC iron-carbon alloy initially containing 0.55 wt% C is exposed to an oxygen-rich and virtually carbon-free atmosphere at 1325 K (1052C). Under these circumstances the carbon diffuses from the...
-
Baxter, Inc., owns 90 percent of Wisconsin, Inc., and 20 percent of Cleveland Company. Wisconsin, in turn, holds 60 percent of Clevelands outstanding stock. No excess amortization resulted from these...
-
An object of mass 2.0 g suspended from the end of a spring has a vibrational frequency of 3.0 Hz. Calculate the force constant of the spring.
-
Calculate the percentage difference in the fundamental vibration wave number of IH35Cl and 2H37CIon the assumption that their force constants are the same.
-
The wave number of the fundamental vibrational transition of 79BrB1Bris 323.2 cm-1, Calculate the force constant of the bond (mC9Br) = 78.9183 u, m(8IBr) = 80.9163 u).
-
The 6 - month LIBOR is 1 . 6 5 % . ( a ) calculate the 6 - month discount factor ( b ) calculate the 6 - month zero - coupon price of a $ 1 bond ( c ) calculate the effective annual yield for that...
-
4. Multi Step Binomial Tree: Consider again the European call option with three months left to maturity written on a non-dividend paying stock. As in exercise 2a, let today's stock price be 100 kr,...
-
C 0 Keenan Industries has a bond outstanding with 25 years to maturity, a 7.45% nominal coupon, semiannual payments, and a $1,000 par value. The bond has a 5.90% nominal yield to maturity, but it can...

Study smarter with the SolutionInn App


