The 1 H NMR spectrum of the most stable isomer of [14]annulene shows two signals, at
Question:
The 1H NMR spectrum of the most stable isomer of [14]annulene shows two signals, at δ = -0.61 (4 H) and 7.88 (10 H) ppm. Two possible structures for [14]annulene are shown here. How do they differ? Which one corresponds to the NMR spectrum described?
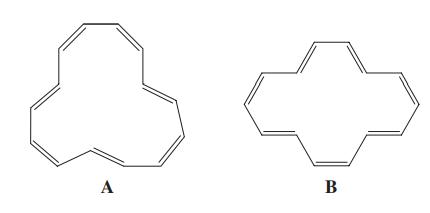
Transcribed Image Text:
A В
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
The two isomers of 14 annulene will interconvert at room temperature but can be obs...View the full answer

Answered By

Vivek Chaudhary
Hi! I am vivek.My greatest passion in life is teaching. I was born and raised in india, and experienced great success at school and at university due to amazing and unforgettable teachers. This is the foundation of my commitment to helping out my students, whatever their abilities may be. Currently, I am studying a masters degree specializing in chemistry, rounding out my undergraduate background in chemistry (Honors) I have been tutoring and teaching for 2 years in various settings – tutoring small and large groups.
So, if you stuck in any problem you can ask.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
The 1H NMR spectrum shown in Figure 14.8 corresponds to one of the following compounds. Which compound is responsible for this spectrum? C CH CH3 CH3 CICH2 CH2C Br2CH CHBr2 6 5 3 2 0 8 (ppm)...
-
The 1H NMR spectrum of 2-propen-1-ol is shown here. Indicate the protons in the molecule that give rise to each of the signals in the spectrum. 8 6 frequency 0
-
The 1H NMR spectrum of compound A (C8H8O) consists of two singlets of equal area at 5.1 (sharp) and 7.2 ppm (broad). On treatment with excess hydrogen bromide, compound A is converted to a single...
-
The following information is computed from Fast Food Chains annual report for 2006. 2006 2005 Current assets $ 2,731,020 $ 2,364,916 Property and equipment, net 10,960,286 8,516,833 Intangible...
-
Explain the skills project managers need to possess in order to have repeated successes in running projects both from a soft skill perspective and technical skill perspective.
-
As of 2024, a company has 1,600 shares of $70 par, 6.0% preferred stock outstanding and 100,000 shares of $1 par common stock outstanding. The preferred stock is cumulative. Preferred stockholders...
-
The following information from Buchanan Company's current operations is available: Required a. Prepare a multiple-step income statement. Disregard earnings per share. b. Prepare a single-step income...
-
The following data were provided by the accounting records of NewFort Limited at year- end, 31 December 20X9: Analysis of selected accounts and transactions: a. Sold plant assets for cash; cost, $...
-
Many large companies have supplier codes of conduct posted on their company websites. Find an example of a supplier code of conduct, post a link to it, and discuss what you find to be the strengths...
-
The lift cage in Figure No2 is hoisted by means of a rope wound round a 0.344 [m] diameter drum. The number of teeths per gear are: Gear Nel is 20. Gear Ne3 is 25, Gear Ne4 is 75. Gear Ne5 is 35....
-
(a) The 1 H NMR spectrum of [18]annulene shows two signals, at = 9.28 (12 H) and -2.99 (6 H) ppm. The negative chemical shift value refers to a resonance upfield (to the right) of (CH 3 ) 4 Si....
-
Explain the following reaction and the indicated stereochemical result mechanistically.
-
It is the end of 2023. Ginnungagap Oy began operations in January 2022. The company is so named because it has no variable costs. All its costs are fixed; they do not vary with output. Ginnungagap is...
-
Identify the items that should be (a) included in the inventory-taking plan and (b) done in observing inventories.
-
For the observation of inventories, indicate (a) when this test is required, (b) the meaning of inventory taking, and (c) the timing and extent of the test.
-
Identify the audit procedures that enable an auditor to verify the (a) existence or occurrence and (b) valuation or allocation objectives for inventory balances.
-
What procedures are useful to the auditor in searching for unrecorded accounts payable?
-
a. What audit procedures are used to verify the existence or occurrence and completeness of accounts payable? b. How may the auditor obtain evidence as to the valuation and completeness of accrued...
-
Zinc has five naturally occurring isotopes: 48.63% of 64 Znwith an atomic weight of 63.929 amu; 27.90% of66Zn with an atomic weight of 65.926 amu; 4.10% of 67Zn with an atomic weight of 66.927 amu;...
-
As long as we can't lose any money, we have a risk-free investment." Discuss this comment. Q2: Both investing and gambling can be defined as "undertaking risk in order to earn a profit." Explain how...
-
Calculate the wavelength of neutrons that have reached thermal equilibrium by collision with a moderator at 300 K.
-
Calculate the lattice enthalpy of Mgbr, from the following data: AH((kJ mol-) Sublimation of Mg(s) +148 Ionization of Mg(g) to Mg+(g) +2187 Vaporization of Br,(1) Dissociation of Br,(g) +31 +193...
-
Sections of the solid fuel rocket boosters of the space shuttle Challenger were sealed together with O-ring rubber seals of circumference 11 m. These seals failed at OC, a temperature well above the...
-
Find the betas of two firms and discuss whether the betas are what you would expect. Then discuss how the returns could be calculated based on the CAPM model. Be sure to state your assumptions.
-
The average daily pay for Tortuga Travel's employees = $589. The adjusting entry to record accrued vacation pay for two days includes: a. a debit to Vacation Benefits Payable = $1,178. b. a credit to...
-
A CPA firm has advertised to look over taxpayers' returns for free. If the CPA firm can get the taxpayer an additional tax refund, the taxpayer and the CPA firm will split the additional refund. Is...

Study smarter with the SolutionInn App


