Explain the following reaction and the indicated stereochemical result mechanistically. H CH3 1. AICI, 2. H,
Question:
Explain the following reaction and the indicated stereochemical result mechanistically.
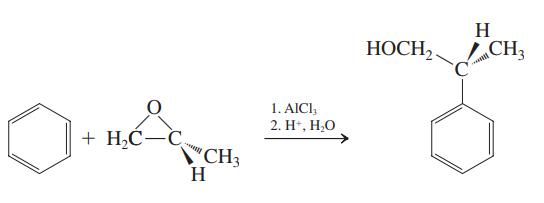
Transcribed Image Text:
H НОСН CH3 1. AICI, 2. H", Н.О + H,C-C CH3 H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
4 HC HC HC 0 Alcl3 H HOHC O A...View the full answer

Answered By

Kalllol Mukherjee
I am a scientist at University of Hyderabad. I have strong background in Chemistry. I have done my B.Sc and M.Sc from Visva-bharati University. I qualified NET with 70 and 34 all India rank in 2013 and 2014 respectively. I have qualified GATE in 2014 with 278 all India rank. Currently I am doing PhD in chemistry from University of Hyderabad. I have published seven research papers in internationally reputed journals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Explain the following reaction sequence. 1. Pd(OCCH,), R,P, K.CO, COCH,CH; Br 2. H,C=CH-COCH.CH,
-
Write a mechanism to explain the following reaction. (As a racemic mixture) IH2O
-
The reaction of an alkenes with bromine in an alcohol as solvent produces as ether as the product. Show a mechanism for the following reaction and explain the stereochemistry of theproduct. Br . H....
-
A small project consists of three activities: A, B, and C. To start activity C, both activities A and B need to be complete. Activity A takes 3 days with a probability of 50 percent and 5 days with a...
-
What were the primary reasons for the growth in project management during the 1990s?
-
Given the table of DELL's Debt-to-equity ratios = Total debt/total shareholders equity Years Total debt Total equity Debt-to-equity ratios 2019 52,056 3155 16.4995 2020 47,984 7,553 6.3530 2021...
-
Notes to the financial statements present information on significant accounting policies, complex or special transactions, details of reported amounts, commitments, contingencies, segments, quarterly...
-
The adjusted trial balance for Karr Farm Corporation at the end of the current year contained the following accounts. Interest Payable $ 9000 Lease Liability 89,500 Bonds Payable, due 2019 180,000...
-
You are the Privacy Office at ABC Hospital you are seeking to enter into a contract with a release of information company. What important elements would you include in the contract (that is, the...
-
A bank is planning to perform efficiency analysis to learn which branch offices are efficient and how to operation of inefficient units can be improved. All the six units performed 10000 transactions...
-
The 1 H NMR spectrum of the most stable isomer of [14]annulene shows two signals, at = -0.61 (4 H) and 7.88 (10 H) ppm. Two possible structures for [14]annulene are shown here. How do they differ?...
-
Metal-substituted benzenes have a long history of use in medicine. Before antibiotics were discovered, phenylarsenic derivatives were the only treatment for a number of diseases. Phenylmercury...
-
Why should young employees be concerned about superannuation?
-
Identify the principal differences between tests of asset balances and tests of current liability balances.
-
In small groups, describe web-based human resources information systems (HRIS) you are familiar with. Based on your own experiences and internet research, evaluate the advantages and disadvantages of...
-
What factors are involved in determining whether a company has been consistent in recording repair and maintenance expenditures?
-
Visit Transparency Internationals website and check to see how the CPI Index for countries listed in Exhibits 5.4 and 5.6 has changed. After searching TIs databank, explain why the changes have...
-
What procedures may be helpful in determining whether all plant asset retirements have been recorded?
-
(a) Calculate %IC of the interatomic bonds for the intermetallic compound Al6Mn? (b) On the basis of this result what type of interatomic bonding would you expect to be found in Al6Mn?
-
A consultant is beginning work on three projects. The expected profits from these projects are $50,000, $72,000, and $40,000. The associated standard deviations are $10,000, $12,000, and $9,000....
-
Young's modulus for iron at room temperature is 215 GPa what strain will be produced when a mass of 10.0 kg is suspended from an iron wire of diameter 0.10 mm?
-
Poisson's ratio for lead is 0.41. What change in volume takes place when a cube of lead of volume 1.0 dm3 is subjected to an uneasily stress that produces a strain of 2.0 per cent?
-
The band gap in silicon is 1.12 eV. Calculate the minimum frequency of electromagnetic radiation that results in promotion of electrons from the valence to the conduction band.
-
If a 25-year ordinary annuity has a present value of 14,643, and an interest rate of 13.0%, what is the amount of each annuity payment to the nearest whole dollar? Question 7 4 pts You are...
-
5. More banks charge an ATM transaction fee for withdrawals than for deposits. What might be the reason for this? 6. If the ABC Bank charges $1.00 per transaction and you use its ATMs 12 times per...
-
eBook Print Item Discussion Question 8-3 (LO. 1) Are land improvements used in a trade or business eligible for cost recovery? Land improvements are eligible for cost recovery, because they have an...

Study smarter with the SolutionInn App


