Write the structures of every enol and enolate ion that can arise from each of the carbonyl
Question:
Write the structures of every enol and enolate ion that can arise from each of the carbonyl compounds illustrated in Problem 32.
Data From Problem 32
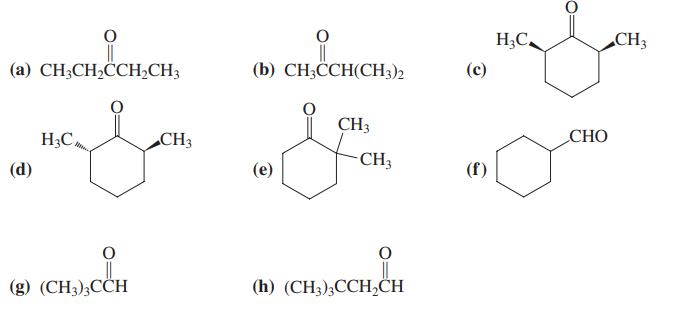
Transcribed Image Text:
H;C. CH3 (a) CH;CH,CH,CH3 (b) CH;CCH(CH3)2 (с) CH3 H;C, CH3 СНО -CH3 (d) (е) (f) (g) (CH3),ССH (h) (CH3),ССH,СH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 38% (13 reviews)

Answered By

Sithara Soman
Worked in an entrance coaching institute as a subject coordinator(Kottakkal , Malappuram-1year). There I have to prepare questions in chemistry based on higher secondary textbooks for students preparing for competitive examinations like NEET,KEAM etc.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Write the structures of the products of reaction of ethylmagnesium bromide, CH3CH2MgBr, with each of the following carbonyl compounds. Identify any reaction that gives more than one stereoisomeric...
-
Write the structure of the enolate ion derived from each of the following -dicarbonyl compounds. Give the three most stable resonance forms of each enolate. (a) 1-Phenyl-1,3-butanedione (b) CH
-
Write the structures of the alkenes that would yield the following carbonyl compounds When treated with ozone and then with dimethyl sulfide. (a) (b) (2 Mol is produced from 1 mol of alkene) (c) o...
-
Adam Kleen Enterprise (AKE) was initially set up as a convenient shop selling laundry related items by Adam Bollan. Despite having good results in the national-level examination and was even offered...
-
This is a case concerning trademark infringement brought by Universal, who owned the rights to the name and character of King Kong, against Nintendo, who produced the famous game, Donkey Kong....
-
Add a function size() to our C++ implementation of a singly link list. Can you design this function so that it runs in O(1) time?
-
1 3 Compare behaviour modification, content and process theories
-
Should Charlies strategy include direct response or confrontation with the negative poster identified by Burris? Look at this! What should we do about it? exclaimed Seymour Burris, Jr., the President...
-
The initial measurement of an acquired item of property plant and equipment is always its cost, unless it is acquired by way of an asset exchange or government grant, in which case the item of...
-
The information listed below refers to the employees of Lemonica Company for the year ended December 31, 2018. The wages are separated into the quarters in which they were paid to the individual...
-
Underline the a-carbons and circle the a-hydrogens in each of the following structures. H;C. CH3 (a) CH;CH,CH,CH, (b) CH,CCH(CH,h (c) CH, H,C. CH3 CHO CH, (d) (e) (g) (CH3);CCH (h) (CH3),CCHCH
-
What product(s) would form if each carbonyl compound in Problem 32 were treated with (a) alkaline D 2 O; (b) 1 equivalent of Br 2 in acetic acid; (c) excess Cl 2 in aqueous base? Data From Problem 32...
-
In 1751, Benjamin Franklin predicted that the U.S. population P(t) would increase with growth constant k = 0.028 year1. According to the census, the U.S. population was 5 million in 1800 and 76...
-
VA= 18/ A c = ? 1. For the arrangement of cable and pulleys in the figure, the velocities and accelerations of two of the bodies involved are indicated. Determine the velocity and acceleration of the...
-
Suppose there are two electric charges in 2D planeR2; one is on y-axis and the other is on x-axis: 45km 0 p1:=0,p2:=30km.(1) Thechargesareq1=4.5Catp1andq2=3.0Catp2. (a) Determine the electric field...
-
6.10 Long Div. and Comp Square Calculus - No Calculator Find the indefinite integral. 1. S 4x-34x+56x-21 4x-2 dx Mastery Check #2 1 dx 2. Sx-4x+5x x-4x+5 S S Name: Sienna Nono Date: 3-1-24 Period:...
-
How well are oncology firms leveraging digital technology to gain and sustain competitive advantage?
-
Axel and Brooklyn have agreed to buy a new vehicle. Brooklyn mentions that she is looking forward to getting a new SUV, so they have room for their dogs and kids. Axel mentions he thought they were...
-
Sabel Co. purchased assembly equipment for $500,000 on January 1, Year 1. Sabels financial condition immediately prior to the purchase is shown in the following horizontal statements model. The...
-
Independent random samples of sizes n1 = 30 and n2 = 50 are taken from two normal populations having the means 1 = 78 and 2 = 75 and the variances 21 = 150 and 22 = 200. Use the results of Exercise...
-
Refer to Fig. 23.3 and determine the pressure range for a branching chain explosion in the hydrogen-oxygen reaction at (a) 700 K, (b) 900 K.
-
Consider the acid-catalyzed reaction Deduce the rate law and show that it can be made independent of the specific term [H+]. (1) HA +H* HAH (2) HAH* +B- BH* + AH k,k, both fast k, slow
-
Consider the following chain mechanism: Use the steady-state approximation to deduce that the rate law for the consumption of A2. (1) A,A+A- (2) A B. + C (3) A- +P B- (4) A-+ B-- P
-
I give likes Rour company uses two measures of activity, routes and passengers in its cost formulas used for budgeting and performance evaluation. The cost formula for tour operating costs is $56,840...
-
Exercise 10-3 (Algo) Variable Overhead Variances [LO10-3] Logistics Solutions provides order fulfillment services for dot.com merchants. The company maintains warehouses that stock items carried by...
-
could you please explain how you got it. Thank you! Aztec Company sells its product for $160 per unit. Its actual and budgeted sales follow. Sales units Sales dollars May (Actual) June (Budget) July...

Study smarter with the SolutionInn App


