A gas mixture contains nitrogen peroxide at 1 atm. The equivalent NO2 is 30% and the dissociated
Question:
A gas mixture contains nitrogen peroxide at 1 atm. The equivalent NO2 is 30% and the dissociated fraction of N2O4 is equal to 0.4. Determine the value for Kp in the equilibrium
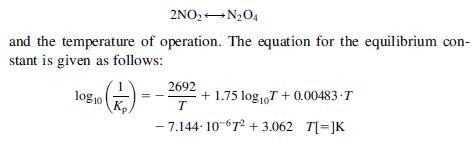
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Industrial Chemical Process Analysis And Design
ISBN: 9780081010938
1st Edition
Authors: Mariano Martín
Question Posted:





