What amount (moles) of compound is present in 1.00 g of each of the compounds in Exercise
Question:
What amount (moles) of compound is present in 1.00 g of each of the compounds in Exercise 53?
Data in Exercise 53?
Calculate the molar mass of the following substances.
a. 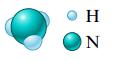
b. 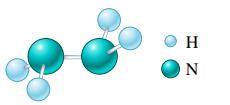
c. (NH4)2Cr2O7
Transcribed Image Text:
Η N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
c NH42Cr2O7 To calculate the molar mass of NH42Cr2O7 we need to sum up the atomic mas...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Calculate the molar mass of the following substances: (a) Li2 CO3, (b) CS2, (c) CHCl3 (chloroform), (d) C6H8O6 (ascorbic acid, or vitamin C), (e) KNO3, (f) Mg3N2.
-
What mass of compound is present in 5.00 moles of each of the compounds in Exercise 54? Data In Exercise 54? Calculate the molar mass of the following substances. a. b. Ca 3 (PO 4 ) 2 c. Na 2 HPO 4 0
-
What mass of compound is present in 5.00 moles of each of the compounds in Exercise 53? Data in Exercise 53? Calculate the molar mass of the following substances. a. b. c. (NH 4 ) 2 Cr 2 O 7 OH N
-
Find the x-values where the following do not have derivatives. + -6 + y 8 0 6 X
-
Use the Holt Winters no trend model to find the best model to find forecasts for the next 12 months in the Excel file Housing Starts.
-
The following data are taken from the records of Cronkite Corp.: Required: Perform horizontal analysis on the above date and interpret your results. 2016 2015 Sales Cost of goods sold Gross profit...
-
In Example 2.2, assume that P.V. Ltd. pays no dividends over its life, until a liquidating dividend is paid at the end of year 2 consisting of its cash on hand at that time. Required Verify that the...
-
Supply the missing information in the following table for Blair Company. Sales ........... $484,000 ROI .......... ? Operating assets ...... ? Operating income ..... ? Turnover ......... 2.2 Residual...
-
Cold Goose Metal Works Inc. is a small firm, and several of itsmanagers are worried about how soon the firm will be able torecover its initial investment from Project Delta??s expected futurecash 2...
-
A government agency has estimated that a flood control project has costs and benefits that are parabolic, according to the equation (Present worth of benefits)2 - 22(Present worth of cost) + 44 = 0...
-
What is the mass of the product? a. Less than 10 g b. Between 20 and 100 g c. Between 100 and 120 g d. Exactly 120 g e. More than 120 g
-
Fig. 3.5 illustrates a schematic diagram of a combustion device used to analyze organic compounds. Given that a certain amount of a compound containing carbon, hydrogen, and oxygen is combusted in...
-
A consumer rights organization wanted to check out different diet plans. It recruited 33 volunteers and sent them to four different programs. After the first 2 weeks, the weight losses, in pounds,...
-
In the text we considered only the zone electrophoresis where a zone of a solute-rich layer is created by the action of an electric field. Other techniques are isotochorphoresis and iso-electric...
-
Various methods have been developed in order to increase the throughput in electrophoresis. Most of these designs vary in the flow arrangement and the changes in the direction of the electric field....
-
The classical Fick or Einstein model for diffusion is based on the assumption that the diffusing molecule is exposed to a random force field arising from molecular motion. The assumption is...
-
The phenomenon of electroosmosis can be used for dewatering and consolidation of soils and mine tailing and waste sludges. The idea is that by appropriate placement of electrodes the flow can be...
-
Reaction in the catalyst pellet with external transport resistances is another classic problem in chemical reaction engineering. This system needs to be modeled as a set of two second-order...
-
Many manufacturers have quality control programs that include inspection of incoming materials for defects. Suppose a computer manufacturer receives circuit boards in batches of five. Two boards are...
-
Which task is performed by a book-keeper? A. Analysing the trading results B. Entering transactions in the ledger C. Preparing year-end financial statements D. Providing information for...
-
This hydroboration reaction forms two products. Show these products and explain which one you expect to be amajor. 1) BH3, THF 2) H,O2. NAOH
-
Show preparation of these alcohols fromalkenes. b) c)
-
Show the products of thesereactions: 1) BH3, THF 2) H2O2, NaOH a) Ph- b) 1) disiamylborane 2) HO2, NaOH
-
Exercise 21-5 (Algo) Preparing flexible budget performance report LO P1 Nina Company prepared the following fixed budget for July using 7,560 units for budgeted sales. Actual sales were 7,260 units...
-
Lab Objectives 0 Use methods ofthe Character class and String class to process text 0 Be able to use the String . spl it method and the StringBuilder class Introduction In this lab we ask the user to...
-
. Menlo Company distributes a single product. The company's sales and expenses for last month follow: Total Per Unit Sales $ 450,000 $ 30 Variable expenses 180, 000 12 Contribution margin 270,000...

Study smarter with the SolutionInn App


