A 0.40-m 3 insulated pistoncylinder device initially contains 1.3 kg of air at 30C. At this state,
Question:
A 0.40-m3 insulated piston–cylinder device initially contains 1.3 kg of air at 30°C. At this state, the piston is free to move. Now air at 500 kPa and 70°C is allowed to enter the cylinder from a supply line until the volume increases by 50 percent. Using constant specific heats at room temperature, determine
(a) The final temperature
(b) The amount of mass that has entered
(c) the work done
(d) the entropy generation.
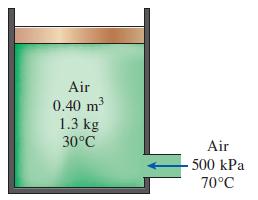
Transcribed Image Text:
Air 0.40 3 1.3 kg m 30°C Air 500 kPa 70°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Air is allowed to enter an insulated pistoncylinder device until the volume of the air increases by ...View the full answer

Answered By

Mwangi Clement
I am a tried and tested custom essay writer with over five years of excellent essay writing. In my years as a custom essay writer, I have completed more than 2,000 custom essays in a diverse set of subjects. When you order essays from me, you are working with one of the best paper writers on the web. One of the most common questions I get from customers is: “can you write my essay?” Upon hearing that request, my goal is to provide the best essays and overall essay help available on the web. I have worked on papers in subjects such as Nursing and Healthcare, English Literature, Sociology, Philosophy, Psychology, Education, Religious Studies, Business, Biological Sciences, Communications and Media, Physical Sciences, Marketing and many others. In these fields, my specialties lie in crafting professional standard custom writings. These include, but are not limited to: research papers, coursework, assignments, term papers, capstone papers, reviews, summaries, critiques, proofreading and editing, and any other college essays.
My extensive custom writings experience has equipped me with a set of skills, research abilities and a broad knowledge base that allows me to navigate diverse paper requirements while keeping my promise of quality. Furthermore, I have also garnered excellent mastery of paper formatting, grammar, and other relevant elements. When a customer asks me to write their essay, I will do my best to provide the best essay writing service possible. I have satisfactorily offered my essay writing services for High School, Diploma, Bachelors, Masters and Ph.D. clients.
I believe quality, affordability, flexibility, and punctuality are the principal reasons as to why I have risen among the best writers on this platform. I deliver 100% original papers that pass all plagiarism check tests (Turnitin, Copyscape, etc.). My rates for all papers are relatively affordable to ensure my clients get quality essay writing services at reasonable prices.
4.50+
5+ Reviews
14+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
A 0.25-m3 insulated pistoncylinder device initially contains 0.7 kg of air at 20°C. At this state, the piston is free to move. Now air at 500 kPa and 70°C is allowed to enter the cylinder...
-
A pistoncylinder device initially contains 1.2 kg of air at 700 kPa and 200°C. At this state, the piston is touching on a pair of stops. The mass of the piston is such that 600- kPa pressure is...
-
A piston-cylinder device initially contains 1.2 kg of air at 700 kPa and 2008C. At this state, the piston is touching on a pair of stops. The mass of the piston is such that 600-kPa pressure is...
-
Explain the difference between an error of commission and an error of principle.
-
What is storage? Why is it risky? What role does it play in the economy? Discuss.
-
Another way to describe a broad range of moods is ________. See if you can fill in the blanks for each of these questions. This exercise is simply to acquaint you with words you might encounter in...
-
What are the components of a global marketing strategy?
-
Rudolph Corporation is evaluating an extra dividend versus a share repurchase. In either case, $11,000 would be spent. Current earnings are $1.40 per share, and the stock currently sells for $58 per...
-
Bergo Bay's accounting system generated the following account balances on December 3 1 . The company s manager knows something is wrong with this list of balances because it does not show any balance...
-
Chuck Wagon Grills, Inc., makes a single producta handmade specialty barbecue grill that it sells for $210. Data for last years operations follow: Required: 1. Assume that the company uses variable...
-
A hot-water pipe at 80C is losing heat to the surrounding air at 5C at a rate of 1600 W. Determine the rate of entropy generation in the surrounding air in W/K.
-
A rigid, adiabatic container is filled through a single opening from a source of working fluid whose properties remain fixed. How does the final specific entropy of the single-phase contents of this...
-
Large samples of women and men are obtained, and the hemoglobin level is measured in each subject. Here is the 95% confidence interval for the difference between the two population means, where the...
-
Using the ideas of kinetic particle theory when you come home from school and open the door you can smell food being cooked
-
The following information relates to Salamat Corporation for the last year.Salamat uses direct labor hours as an overhead base. Estimated direct labor hours 360,000 hours Estimated manufacturing...
-
Code in matlab the translational motion via numeric integration of the orbit (two-body orbit sufficient). Use the orbital characteristics of the Centaur V upper stage from the Atlas V launch on...
-
Lolita Company has the following information available for June 2020: Beginning Work in Process Inventory (25% as to conversion) 20,000 units Started 130,000 units Ending Work in Process Inventory...
-
Question 3 (15 marks) Sporty Ltd. produces scooters and skateboards. At the beginning of the year, the following volume of activities were budgeted for the year: Production volume/units Direct labour...
-
An interaction effect in the model from a factorial experiment involving quantitative factors is a way of incorporating curvature into the response surface model representation of the results. True...
-
5. How much would you need to deposit in an account now in order to have $5,000 in the account in 5 years? Assume the account earns 2% interest compounded monthly. 10. You deposit $300 each month...
-
An inventor claims to have devised a cyclical engine for use in space vehicles that operates with a nuclear-fuel-generated energy source whose temperature is 920 R and a sink at 490 R that radiates...
-
A heat engine receives heat from a heat source at 1200C and has a thermal efficiency of 40 percent. The heat engine does maximum work equal to 500 kJ. Determine the heat supplied to the heat engine...
-
In tropical climates, the water near the surface of the ocean remains warm throughout the year as a result of solar energy absorption. In the deeper parts of the ocean, however, the water remains at...
-
Series of Compound Interest Techniques The following are several situations involving compound interest. Required: Using the appropriate table, solve each of the following: ( Click here to access the...
-
If Clark Kelly has recognized gain on an exchange of like-kind property held for investment use, where does Clark report the gain? First on Form 8824, then carried to Schedule D. First on Form 8824,...
-
An investor put 40% of her money in Stock A and 60% in Stock B. Stock A has a beta of 1.2 and Stock B has a beta of 1.6. If the risk-free rate is 5% and the expected return on the market is 12%,...

Study smarter with the SolutionInn App


