The herbicide oxyfluorfen can be prepared by reaction between a phenol and an aryl fluoride. Propose amechanism.
Question:
The herbicide oxyfluorfen can be prepared by reaction between a phenol and an aryl fluoride. Propose amechanism.
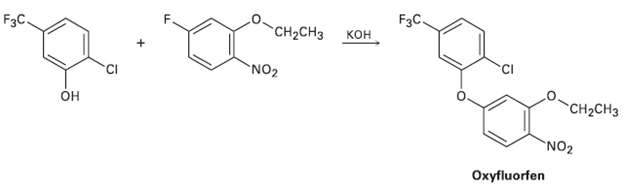
Transcribed Image Text:
FзC. кон F3C. "CH-CHз NO2 он "CH-CHз NO2 Охyfluorfen
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (19 reviews)
Hydroxide is used to form the nucleophilic phenoxide anion F3C F3C F...View the full answer

Answered By

Kennedy Odhiambo
As a professional writer, I have been in the field for over 5 years having worked as a lecture in different tertiary institutions across the world. With this impeccable experience, I assure provision of a good and supporting environment for students to learn.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following alkyl bromide can be prepared by reaction of the alcohol (S)-2-pentanol with PBr3. Name the compound, assign (R) or (S) stereochemistry, and tell whether the reaction of the alcohol...
-
The herbicide acifluorfen can be prepared by a route that begins with reaction between a phenol and an aryl fluoride. Propose a mechanism. NO2 CO2CH3 NO2 co,CH3 NO2 .CO2H DMSO HO F3C F3C CI FC...
-
A compound known as Hagemann's ester can be prepared by treating a mixture of formaldehyde and ethyl acetoacetate first with base and then with acid and heat. Write the structure for the product of...
-
Calculate e y for the following values of y: (a) y = 1 (b) y = 5 (c) y = -.5 (d) y = -2.5 (e) y = 3.1 (f) y = - I (g) y = .05 (h) y = .32 (i) y = 6.1 (j) y = -5.4
-
Lonzo owns two apartment buildings. He acquired Forsythia Acres on February 21, 1998, for $300,000 ($90,000 allocated to the land) and Square One on November 12, 2016, for $800,000 ($100,000...
-
Over the past few years the percentage of students who leave Lakeland College at the end of the first year has increased. Last year Lakeland started a voluntary one-week orientation program to help...
-
A \(2 \mathrm{~cm}\)-diameter, \(19 \mathrm{~cm}\)-long tube is placed touching a pool of liquid. The end away from the liquid pool \((\mathrm{z}=0.19 \mathrm{~m})\) is in an air stream (component C)...
-
Merger Valuation with Change in Capital Structure VolWorld Communications Inc., a large telecommunications company, is evaluating the possible acquisition of Bulldog Cable Company (BCC), a regional...
-
Measuring stand - alone risk using realized ( historical ) data Returns earned over a given time period are called realized returns. Historical data on realized returns is often used to estimate...
-
The dating web site Oollama.com requires its users to create profiles based on a survey in which they rate their interest (on a scale from 0 to 3) in five categories: physical fitness, music,...
-
Show the major product(s) from reaction of the following substances with (i) CH3CH2Cl, AlCl3 and (ii) HNO3,H2SO4 (b) (a)
-
Treatment of p-bromotoluene with NaOH at 300 oC yields mixture of two products but treatment of m-bromotoluene with NaOH yields a mixture of three products. Explain.
-
Which of the following items is reported as a current liability on the balance sheet? a. Short-term notes payable b. Estimated warranties c. Payroll withholdings d. All of the above
-
Discuss the three models of the role of IT in change and what each role implies for the IT manager. What do the roles suggest for IS strategy? What might determine which model is most appropriate in...
-
Do you agree or disagree with the assumption that culture is an important impediment (or facilitator) of effective IT implementation? What are some situations you have experienced that confirm or...
-
Give some examples that support the view of technology determinism (the ability of technology, as opposed to people, to cause change). What are some contravening examples?
-
If the propositions in the chapter are correct, what are the implications, if any, for organizational performance?
-
What are the implications of these key issues for small-medium sized local firms?
-
In a uniaxial state of stress, the maximum shear stress occurs on a plane which is inclined to the load line at (a) \(0^{\circ}\) (b) \(45^{\circ}\) (c) \(60^{\circ}\) (d) \(90^{\circ}\)
-
After graduating from college and working a few years at a small technology firm. Preet scored a high-level job in the logistics department at Amex Corporation. Amex sells high-quality electronic...
-
By measuring the temperature change accompanying a differential volume change in a free expansion across a valve and separately in a reversible adiabatic expansion, the two derivatives (T/ V ) H and...
-
Explain which mechanism is preferred in these reactions and show the majorproducts: Br b) 1-BUOH 0- PROH Br . EIOH, reflux OTs d) DMF + CH,CO + KOH Ph Br Br . ELOH ELOH f) e) + CH,CH,0
-
Explain which reaction mechanism (E1, SN1, E2, SN2) these reactions follow, and show the majorproducts: Br a) DMSO + CN Br b) . reflux + OH Br CH c) EIO LOTS + NaOEt d) t-BUOH CI + t-BUOK e) . Br CH,...
-
Show all the steps in the mechanism for this reaction. What substitution product(s) would also be formed in thisreaction? Br E:OH
-
Suppose your firm is considering investing in a project with the cash flows shown below, that the required rate of return on projects of this risk class is 11 percent, and that the maximum allowable...
-
Suppose we wish to lend $1,000,000 for 91 days at SOFR (the successor to LIBOR) beginning next June. In this case, we might want to hedge against a potential interest rates between now and June by...
-
The next TWO questions are based on the following information. Nadia and Robert want to buy a house. They have a combined gross income of $72,000, and can apply $50,000 towards the downpayment of a...

Study smarter with the SolutionInn App


