Will either of the following reactions take place as written, according to the data inTable? (a) HCN
Question:
Will either of the following reactions take place as written, according to the data inTable?
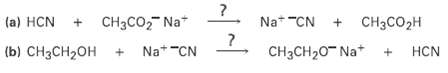
Transcribed Image Text:
(a) HCN + CH3CO2 Na+ Na* "CN CHзCO2H Na* "CN CH3CH20- Na* |(b) CH3CH2OH HCN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Strategy Use Table to find the strength of each acid A reaction take...View the full answer

Answered By

PRINCE PANDEY
I am Indian Chartered Accounting having a strong hold in the subjects of Accounting, IFRS Reporting, Indian
Taxation, Cost Accounting, Auditing. I have vast experience of teaching a student with easy way problem-solving approach.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following reactions take place in a batch reactor: A + B ( C (desired product) B + C ( D (hazardous product) As the reaction proceeds, D builds up in the reactor and could cause an explosion if...
-
The enthalpy change for each of the following reactions was calculated using bond energies. The bond energies of XO, YO, and ZO are all equal. XX + O=O XOOX; H = 275 kJ YY + O=O YOOY; H = +275 kJ...
-
As discussed in Section 6.2, either of the following two equations can be applied to determine the amount necessary to recover invested capital and a required return: CR 1 = P(AP,i,n) + S(AF,i,n) or...
-
Differentiate between formal and behavioral roles, and describe how behavioral roles emerge during group interaction.
-
Melissa owns a residential lot in Spring Creek, Louisiana, that has appreciated substantially in value. She holds the lot for investment. She is considering exchanging the lot for a residential lot...
-
Compare and contrast check boxes and radio buttons. When would you use one versus the other?
-
The year 2000 was not a particularly pleasant year for the managers of Xerox Corporation, or its shareholders. The company's stock price had already fallen in the previous year from $60 per share to...
-
Robert, a new client of yours, is a self-employed caterer in Santa Fe, New Mexico. Robert drives his personal van when delivering catered meals to customers. You have asked him to provide the amount...
-
Proceeds from Notes Payable On January 2 6 , Nyree Co . borrowed cash from Conrad Bank by issuing a 4 5 - day note with a face amount of $ 2 2 5 , 0 0 0 . Assume a 3 6 0 - day year. a . Determine the...
-
Let S= {a,b,c,d,e} and P be the set of partitions of S such that P={P1,P2,P3,P4}, where P1={{a,b,c},{d,e}},P2={{a,b},{c,d,e}},P3={{a,b,c,d,e}} and P4= {{a},{b},{c},{d},{e}} A partial order is defined...
-
Amide ion, H 2 N , is a stronger base then hydroxide ion, HO . Which is the stronger acid, NH 3 or H 2 O? Explain.
-
Ammonia, NH3, has pKa = 36 and acetone has pKa 19. Will the following reaction takeplace? Na* -:NH2 NH3 CH H Acetone Na+ CH2:
-
Use the stepwise regression procedure to find the best model to describe miles per gallon for the data in Table 10. Use a level of significance of \(\alpha=0.15\) as the criteria for an explanatory...
-
Kunkel Company is considering the purchase of a $25,000 machine that would reduce operating costs by $6,000 per year. At the end of the machine's five-year useful life, it will have zero salvage...
-
Company Z just paid an annual dividend of $1.22 a share. The stock has a market value of $34.60 and a dividend growth rate of 3.1 percent. What is the rate of return on this stock?
-
Should companies be responsible for fostering a work-life balance for their employees or is it solely the responsibility of the individual employee? Please explain your position.
-
I'm researching topics concerning Current Or Emerging Technology Ethics. Here is the following: 1. Papers Objective . The objective of this paper and outline is to: Evaluate a new ethical issue (not...
-
A. Evatuate the two divergent challenges faced by Microfinance institutions(MFIs). Identify and explain the measurements used to assess the performance of MFIs in respect to these two challenges....
-
Write the section of code that would display a list of services in a Label object named lb1Service if the user selects any of the corresponding CheckBoxes shown in Figure 9-78. chkOutside Wash Car...
-
Match each of the key terms with the definition that best fits it. _______________ A record of the sequence of data entries and the date of those entries. Here are the key terms from the chapter. The...
-
a. Calculate the fugacity of liquid hydrogen sulfide in contact with its saturated vapor at 25.5C and 20 bar. The vapor pressure of pure water at 310.6 K is 6.455 kPa. Compute the fugacity of pure...
-
Name these compounds: CH3 CH3 Br a) CHCHCHC-CHCHCH3 CH3 b)
-
Draw the structure of 3-bromo-4-butylcyclohexene.
-
Name these compounds: a) CHCHCHCH3 c) -CHCHCH b) CHCHCCHCHCH CH d) Br CH3
-
A company has fixed costs of $250,000, variable costs per unit of $5, and a selling price per unit of $18. Calculate the breakeven point in units and dollars, and evaluate the company's cost...
-
The balance sheet of XYZ Ltd. on December 31, 2023, is provided below: Item Amount ($) Accounts Payable 1,230,000 Notes Payable 1,210,000 Accrued Expenses 1,090,000 Total Current Liabilities ?...
-
Calculate Debt to Equity Ratio: Requirements: Given the balance sheet information: Total Debt: $300,000 Total Equity: $450,000 Compute the debt to equity ratio. Express the debt to equity ratio as a...

Study smarter with the SolutionInn App


