For each of the following compounds, determine the position that is most likely to be the site
Question:
(a)
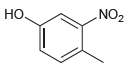
(b)
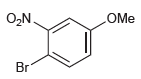
(c)
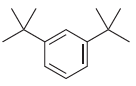
(d)
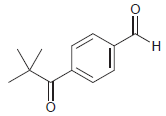
(e)
Transcribed Image Text:
Но. NO2 O,N OMe Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
a b...View the full answer

Answered By

BRIAN MUSINGA
I possess a Bachelors of Commerce degree(Marketing option) and am currently undertaking an MBA in marketing. I believe that I possess the required knowledge and skills to tutor in the subject named. I have also written numerous research academic papers much to the satisfaction of clients and my professors.
5.00+
2+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds, compare the two indicated protons and determine whether they are enantiotopic, homotopic, or diastereotopic: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m)...
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) Discuss. OMe . CI H,
-
For each of the following compounds determine which (if any) lone pairs are participating in aromaticity. a. b. c. d. e. f. g. h. :N-H
-
Consider the plane stress inhomogeneous case with only variation in elastic modulus given by E = E(y) = 1/ (Ay + B). Further assume that the Airy function depends only on y, = (y). Show that...
-
Describe how a small business owner could use the following sources of a competitive advantage: "niche-picking," "entertailing," emphasizing their uniqueness, connecting with their customers,...
-
(a) Find the matrix in R3 that corresponds to a counterclockwise rotation around the x-axis through an angle 60. (b) Write it as a product of elementary matrices, and interpret each of the factors.
-
Use the Convolution Theorem to compute the inverse transform of the following: a. \(F(s)=\frac{2}{s^{2}\left(s^{2}+1 ight)}\). b. \(F(s)=\frac{e^{-3 s}}{s^{2}}\). c. \(F(s)=\frac{1}{s\left(s^{2}+2...
-
The Starr Theater, owned by Meg Vargo, will begin operations in March. The Starr will be unique in that it will show only triple features of sequential theme movies. As of March 1, the ledger of...
-
. Marius Boring, the senior partner of the auditing firm of Dull and Void, has had a car accident, as a result of which he cannot remember anything about auditing since Enron. He thus needs to be...
-
Mr. Chai has been trading for some years as a wine merchant. The following list of balances has been extracted from his ledger as at 30 April 20X7, the end of his most recent financial year. Ghc...
-
When 2,4-dibromo-3-methyltoluene is treated with bromine in the presence of iron (Fe), a compound with molecular formula C8H7Br3 is obtained. Identify the structure of this product.
-
For the reaction C(graphite) + H 2 O(g) CO(g) + H 2 (g), H o R =131.28 kJ mol -1 at 298.15 K. Use the values of C P,m at 298.15 K in the data tables to calculate H R at 125.0C.
-
Organizational strategy can be divided roughly into two categories: a) formulation and b) implementation. Although there is legitimate crossover between the two, how would you characterize the issues...
-
Jane is considering a 7/23 balloon mortgage with an interest rate of 4.15% to purchase a house for $197,000. What will be her monthly payment for the first 7 years of the balloon mortgage?
-
(1) Consider a low-pressure chemical vapor deposition diffusion reactor to lay a thin film of the semiconductor gallium, Ga, onto a silicon wafer surface. Gallium metal is not volatile, but trimethyl...
-
Suppose the exchange rate between U.S. dollars and Swiss francs is SF 1.112 = $1.00, and the exchange rate between the U.S. dollar and the euro is $1.00 = 0.9842 euros. What is the cross-rate of...
-
Assume B Corporation has $375,000 of Ordinary Income from its operations, $75,000 of interest received from its investments on bonds from Z firm. Also, it received $50,000 of dividends from a...
-
1. 2. 3. 7 5. 7. 8. 9. 10. Name the two services that are part of the System Manager. List and describe each of the features found in these services. What is the purpose of identifying account...
-
Go to the home pages of the following retail trade publications: Chain Store Age at www.chainstoreage.com, and Retailing Today at www.retailingtoday.com. Find an article in each that focuses on...
-
Wal-Mart is the second largest retailer in the world. The data file on the disk holds monthly data on Wal-Marts revenue, along with several possibly related economic variables. a) Using computer...
-
Use Figure 12.2 to answer the following question. How does the energy of X-rays compare with that of blue light (greater or smaller)? Figure 12.2 1023 02cosmic rays 1oto 10g 108 107 106 10* 104 10...
-
(a) You have found in the laboratory two liquids, C and D, in unlabeled bottles. You suspect that one is deuterated chloroform (CDC13) and the other is ordinary chloroform (CHC13). Unfortunately, the...
-
(a) You have found in the laboratory two liquids, C and D, in unlabeled bottles. You suspect that one is deuterated chloroform (CDC13) and the other is ordinary chloroform (CHC13). Unfortunately, the...
-
The following companies have their financial year ended on 31 December 2021.1. Entity 1 has 1,000,000 ordinary shares at 1 each as on 1 January 2021. On 1 April 2021, the company issued 250,000 ord 2...
-
Coco Ltd. (Coco) purchased 90% of the voting shares of FerdinandInc. (Ferdinand) for $612,000 on January 1, 2022. On that date,Ferdinand's common shares and retained earnings were valued at$200,000 2...
-
. m 7) The program should be well-documented(comments) and easy to read. Bonus feature (Optional) 1) Allow the user to specify delivery ($5) or pickup ($10) and calculate the appropriate cost. The...

Study smarter with the SolutionInn App


