In Chapter 21, we will explore how nitriles can be converted into carboxylic acids. How would you
Question:
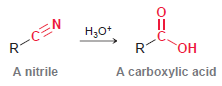
Transcribed Image Text:
R-CEN A nitrile Н,о* C. он A carboxylic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The starting material has a cyano group C N and is ...View the full answer

Answered By

Lisper Wanja
I am an experienced and highly motivated writer with a passion for the skills listed. I have a proven track record of my expertise and my aim is to deliver quality, well-detailed and plagiarism free projects. My genuine passion for writing combined with my ongoing professional development through school and research makes me an ideal candidate within for any assignment.
4.90+
233+ Reviews
388+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would you use IR spectroscopy to distinguish between the following pairs of compounds? (a) (b) N.
-
A tert-l3utyI esters [RCO2C (CH3)3] are converted into carboxylic acids (RCO2H) by reaction with trifluoroacetic acid, a reaction useful in protein synthesis (Section 26.7). Assign E, Z designation...
-
Show how cyclohexylacetylene can be converted into each of the following compounds: a. b.
-
Because Natalie has been so successful operating Cookie Creations, Katy would like to have Natalie become her partner. Katy believes that together they will create a thriving cookie-making business....
-
What appears to be the current operating target of the Fed?
-
Estimate the (approximate maximum) horsepower needed to accomplish the operation described in Problem 2 at a cutting speed of 10 m/min. (Hint: First find the HP used per tooth and determine the...
-
Use your calculator to calculate \(-750 /(-3)\) Explain how the answer agrees with what was expected.
-
Hobart, Evans, and Nix is a small law firm that employs 10 partners and 12 support persons. The firm uses a job-order costing system to accumulate costs chargeable to each client, and it is organized...
-
The following transactions were completed by Winklevoss Inc, whose fiscal year is the calendar year Year 1 July Oct. 1 Issued $76,600,000 of 20-year, 8% callable bonds dated July 1, Year 1, at a...
-
On July 29, 2012, Ever Green Company, a garden retailer, purchased $12,000 of seed, terms 2/10, n/30, from Fleck Seed Co. Even though the discount period had expired, Mary Jasper subtracted the...
-
If one country determines it wants a fixed exchange rate with another a. It can do nothing on its own, it must have the cooperation of the other country. b. It only needs to announce its desired...
-
For tax purposes, gross income is all the money a person receives in a given year from any source. But income taxes are levied on taxable income rather than gross income. The difference between the...
-
Fill in the blanks with an appropriate word, phrase, or symbol(s). When using lattice multiplication, after using multiplication to complete the boxes in the rectangle, add the numbers along the...
-
What functions are involved in the processing of cash receipts transactions?
-
a. Describe the types of computer service organizations that may be used by an entity. b. Indicate the effects of service organizations on the auditor's consideration of the ICS.
-
Vouching recorded sales to supporting documents is a test often performed by auditors as a dual purpose test. Explain.
-
What knowledge of the EDP system should the auditor acquire in obtaining an understanding?
-
What cutoff tests are performed for accounts receivable, how are they performed, and to what account balance audit objectives does the evidence pertain?
-
Whats the electric field strength in a region where the flux through a 1.0 cm x 1.0 cm flat surface is 65 Nm 2 /C, if the field is uniform and the surface is at right angles to the field?
-
Write an essay describing the differing approaches of nursing leaders and managers to issues in practice. To complete this assignment, do the following: 1. Select an issue from the following list:...
-
Name the followingcompounds: CH (b) (c) SH (a) CH SH CH CH3CH2CHSH CHH-CHH2H CH (e) C (d) (f) SCH3 CHCHSCH2CH3 SCH3 SCH2CH3
-
2-Butene-1-thiol is one component of shunk spray. How would you synthesize this substance from methyl 2-butenoate? From 1,3-butadiene CH3CH=CHCOCH3 CH3CH=CHCH2SH 2-Butene-1-thiol Methyl 2-butenoate
-
The 1H NMR spectrum shown is that of an ether with the formula C4H8O. Propose astructure. TMS 10 9. 8. 7. 6. O ppm Chemical shift (8) Intensity 3.
-
1. Sharing buffers to handle multiple sources of variability is referred to a. buffering b. overflow c. concentrator d. pooling 2. A cost-effective approach to reducing buffering costs in a...
-
For this assignment identify on example of abuse of power. This abuse of power can pertain to corporate, religious or other organizational environment (Example: Enron) Then research three (3)...
-
Foreign direct investment (FDI) occurs a.Both when there is an acquisitionby a foreign entity in the U.S., of 10 percent or more of the voting shares of a business ANDwhen an investor acquires a...

Study smarter with the SolutionInn App


