Acetanilide is less reactive than aniline toward electrophilic substitution.Explain. c%3D0 Acetanilide CH
Question:
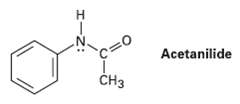
Acetanilide is less reactive than aniline toward electrophilic substitution.Explain.
Transcribed Image Text:
н c%3D0 Acetanilide CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (18 reviews)
08 II CO CH3 H C8 08 co CH3 0 00 CH3 to y less favored CH3 08 ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which compound is more reactive toward electrophilic substitution (for example, nitration)? OCH or b. CH,CH3 a. ETor
-
Explain why phenoxide ion undergoes electrophilic aromatic substitution even more easily than does phenol.
-
Alkenes are more reactive than alkynes toward addition of electrophilic reagents (i.e., Br2, Cl2, or HCl). Yet when alkynes are treated with one molar equivalent of these same electrophilic reagents,...
-
The monthly earnings of financial analysts are normally distributed with a mean of $5,700. If only 6.68 % of the financial analysts have a monthly income of more than $6,140, what is the value of the...
-
Wolfgang, who is age 33, records AGI of $125,000. He incurs the following itemized deductions for 2016. Medical expenses [$15,000 - (10% $125,000)].................................$ 2,500 State...
-
What are fixed assets? Why is assessing risk important in capital budgeting?
-
Sam Weber finances a new automobile by paying $6,500 cash and agreeing to make 40 monthly payments of $500 each, the first payment to be made one month after the purchase. The loan bears interest at...
-
Hayley Doll owns a car stereo store. She has divided her store into three departments. Net sales for the month of July are as follows: Deluxe: ........$33,600 Standard: ........ 38,400 Economy:...
-
The Managing Director of a Trading Company, has just received summary sets of Financial Statements for last year and this year. Trading Company Income Statements for years ended December 31 last year...
-
A rocket is shot straight up from the earth, with a net acceleration (= acceleration by the rocket engine minus gravitational pullback) of 7t m/sec 2 during the initial stage of flight until the...
-
An electrostatic potential map of (trifluoromethyl) benzene, C6H5CF3, is shown. Would you expect (trifluoromethyl) benzene to be more reactive or less reactive than toluene toward electrophilic...
-
Draw resonance structures for the intermediates from reaction of an electrophile at the ortho, Meta and Para positions of nitrobenzene. Which intermediates are most stable?
-
A 20-cm-diameter sphere is totally enclosed by a large mass of glass wool. A heater inside the sphere maintains its outer surface temperature at 170C while the temperature at the outer edge of the...
-
Let $N$ be a positive integer. Consider the relation $\circledast$ among pairs of integers $r, s \in \mathbb{Z}$ defined as $r \circledast s$ when $r-s$ is an integer multiple of $N$. Prove that...
-
Draw a circuit diagram for a typical home hair dryer. To which form (or forms) of energy is electric potential energy converted when you use the dryer?
-
Draw a vector field diagram for particles carrying charges \(+2 q\) and \(-q\) separated by a distance \(r\). Comment on the significance of the vector diagram.
-
(a) Show that the Jones matrix of a polarization analyzer set at angle \(\alpha\) to the \(X\)-axis is given by \[ \underline{\mathbf{L}}(\alpha)=\left[\begin{array}{cc} \cos ^{2} \alpha & \sin...
-
Let \(\mathbf{V}(t)\) be a linearly filtered complex-valued, wide-sense stationary random process with sample functions given by \[ \mathbf{v}(t)=\int_{-\infty}^{\infty} \mathbf{h}(t-\tau)...
-
How is an organizations culture formed and sustained?
-
In Problem 8.43, determine the smallest value of for which the rod will not fall out of the pipe. IA -3 in.-
-
The speed of propagation of a small pressure pulse or sound wave in a fluid, v S , can be shown to be equal to where is the molar density. a. Show that an alternative expression for the sonic...
-
Both cis and trans alkenes can be formed from this compound y anti elimination. Draw a Newman projection of the conformation required to form each of these products and, on the basis of these...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
Explain why one of these compounds reacts readily by an E2 mechanism when treated with sodium ethoxide in ethanol but the other doesnot: OTs OTS . H, .H, CH; CH3
-
QUESTION 3 A business owns seven flats rented out to staff at R500 per month. All flats were tenanted Ist january 21 months rent was in arrears and as at 31st December 14 months' rent wa Identify the...
-
1. 2. 3. Select the Tables sheet, select cells A6:B10, and create range names using the Create from Selection button [Formulas tab, Defined Names group]. Select cells B1:F2 and click the Name box....
-
Tropical Rainwear issues 3,000 shares of its $18 par value preferred stock for cash at $20 per share. Record the issuance of the preferred shares. (If no entry is required for a particular...

Study smarter with the SolutionInn App


