Aromatic compounds such as benzene react with alkyl chlorides in the presence of A1C1 3 catalyst to
Question:
Aromatic compounds such as benzene react with alkyl chlorides in the presence of A1C13 catalyst to yield alkylbenzenes. The reaction occurs through a carbocation intermediate, formed by reaction of the alkyl chloride with A1C13 (R-C1 + AlCl3 ? R+ + A1C14-). How can you explain the observation that reaction of benzene with 1-chioropropane yields isopropylbenzene as the major product?
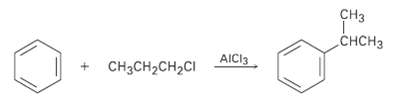
Transcribed Image Text:
сз CHCH3 AICI3 CнзCH-CH2CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Reaction of 1chloropropane with the Lewis acid AlCl3 forms a ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Alkyl halides add to alkenes in the presence of AlCl3; yields are the highest when tertiary halides are used. Predict the outcome of the reaction of tert-pentyl chloride (1-chloro-2,...
-
In the presence of the enzyme aconitase, the double bond of aconitic acid undergoes hydration. The reaction is reversible, and the following equilibrium is established:
-
In the presence of hidden action problems, under what conditions will a deferred compensation contract both minimize taxes and provide desirable work incentives for employees?
-
Conduct some additional research to learn more about Fabletics. How is Fabletics meeting customer needs through its value delivery network? What controversy surrounds the company? What type of...
-
Identify a business or sports leader who you think is highly effective. Present your observations to the class.
-
Repeat the preceding problem assuming that m(t) is the triangular pulse 4Î[1/3(t - 6)]. Data From Problem 11 An FM modulator has output where f d = 20 Hz/V. Assume that m(t) is the rectangular...
-
48. Jarvie loves to bike. In fact, he has always turned down better-paying jobs to work in bicycle shops where he gets an employee discount. At Jarvies current shop, Bad Dog Cycles, each employee is...
-
The following financial data were reported by 3M Company for 2006 and 2007 (dollars in millions). Instructions(a) Calculate the current ratio and working capital for 3M for 2006 and 2007.(b) Suppose...
-
1. Studies findi that entrepreneurs have higher rates of savings relative to other groups in society. Outline some reasons to explain this. 2, What is the private equity premium puzzle that relates...
-
7. On 2 January 2019, the company obtained for free from the Malaysian government a license that allows the company to export goods to overseas for an indefinite period. On that date, the fair value...
-
Epi-Aristolochene, a hydrocarbon found in both pepper and tobacco, is biosynthesized by the following pathway. Add curved arrows to show the mechanism of each step. Which steps involve alkene...
-
Alkenes can be converted into alcohols by acid-catalyzed addition of water. Assuming that Markovnikov?s rule is valid, predict the major alcohol product from each of the following alkenes. H CH3CH-CH...
-
Mortgage lenders often use FICO scores to check the credit worthiness of consumers applying for real estate loans. In general, FICO scores range from 300 to 850 with higher scores representing a...
-
Machine cost = $15,000; life = 8 years; salvage value = $3,000. What minimum cash return would an investor demand annually from the operation of this machine if he desires interest annually at the...
-
Write a program that prompts for the student's name, the number of exams, the exam score of each exam, and display the letter grade for the student. Read the entire problem description before coding....
-
Considering only the vertical stabilizer and rudder, explain the aerodynamic forces and moments that are created. You must include at least applicable airfoil terminology, description of force...
-
part. Review A bicycle wheel is rotating at 47 rpm when the cyclist begins to pedal harder, giving the wheel a constant angular acceleration of 0.44 rad/s. Part B How many revolutions does the wheel...
-
Suppose the number of students who register for a certain class each semester can be modeled by a Poisson distribution with average 10. Suppose further that each student passes the class with...
-
Physical measures do not illustrate the impact on net income of variations from standard. LO.1
-
Suppose you are comparing just two means. Among the possible statistics you could use is the difference in means, the MAD, or the max min (the difference between the largest mean and the smallest...
-
What happens to a system in dynamic equilibrium when it is disturbed in some way?
-
Draw the more stable chair conformation for each of the following compounds: CI Cl Cl
-
Deterrtine whether each of the following compounds can in principle be isolated in optically active form under ordinary conditions. (a) 1.1 -dimethyl cyclohexane (b) cis- l -ethyl-3-methylcyclohexane
-
Draw a structure for the more stable conformation on of tmns-1,2-dimetrylcyclobutane.
-
Eye Deal Optometry leased vision - testing equipment from Insight Machines on January 1 , 2 0 2 4 . Insight Machines manufactured the equipment at a cost of $ 2 0 0 , 0 0 0 and lists a cash selling...
-
help! ee all photos + Add to o e D C N X Edit & Create Share Table of Contents No sales to an individual customer accounted for more than 10% of revenue during any of the last three fiscal years. Net...
-
Business law A person may have the liability of a partner even though no partnership exists True False

Study smarter with the SolutionInn App


