Assign the configurations of these compounds as Z or E: 3 CH,CH2CH3 C=C CH,Br C=C HOCH,CH2 b)
Question:
Assign the configurations of these compounds as Z or E:
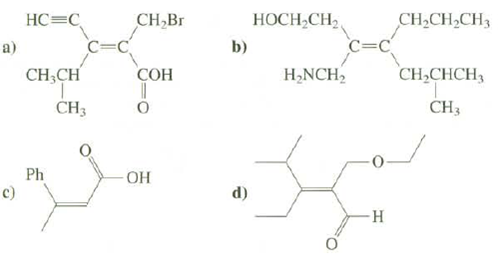
Transcribed Image Text:
НС3С CH,CH2CH3 C=C CH,Br C=C HOCH,CH2 b) a) H,NCH, CH,CHCH3 CH,CH СОН CH3 ČH3 - HO- Ph c) d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 93% (16 reviews)
a Z ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign the configurations of the compounds represented by these Fischer projections as R or S. CH -CH2CH3 b) CH2=CH- ) >N- CH-OH CH3 Serine
-
Assign the configuration of these compounds as R or S: a) c) e) CH3 HC C HOCH H HC g) H- B CH CH3 i) HN- -OH CH H CH3 COH FH H CH,Ph Phenylalanine b) d) f) Br CH3 "CO,H h) H- me H to Br OH -CH3...
-
Assign these compounds as the Z or Eisomers: a) HC C-C CHCH2 CHCI CH3 N OH b) H F C=C C=CH CHCH
-
Carter Co. has a value of $80 million. Buleigh is otherwise identical to Carter Co., but has $32 million in debt. Suppose that both firms are growing at a rate of 5%, the corporate tax rate is 38%,...
-
Porter Ltd began operation on 1 January, and achieved the following results for the year: Sales .................................................36 000 units Selling price...
-
Describe the basic structure of secondary markets. Be sure to differentiate between broker markets and dealer markets.
-
The Manhattan Company has \($265\) million in assets, \($90\) million in current liabilities, \($135\) million in noncurrent liabilities, and \($40\) million in shareholders equity. Of the companys...
-
The portfolio manager of a hedge fund believes that stock A is undervalued and stock B is overvalued. Currently their prices are $30 and $30, respectively. The portfolio manager of the fund buys 100...
-
II. An electric supplies commerce sells some devices 80.000 units a year for $130 and the cost of unit is $126 in general. Research on sales quantity showed that a 40% increase in production quantity...
-
Internet Consulting Service, Inc., adjusts its accounts every month. The company's year-end unad- justed trial balance dated December 31, 2018 follows. (Bear in mind that adjusting entries already...
-
Which of these groups has the higher priority? Br b) -C=N or -CH,CH2 a) -C=CH or CH3 CH3 c) -C=CH2 or -C-CH3 CH3 -C-CH3 d) -C=CH or CH3
-
How would the energy versus dihedral angle plot for 2-methylpropane (isobutene) differs from that for propane?
-
The following information relates to Armanda Co. for the year 2022. After analyzing the data, prepare an income statement and an owner?s equity statement for the year ending December 31, 2022....
-
a. The aging of Torme Designs' accounts receivable is shown below. Complete the schedule and determine the total estimate of uncollectible accounts. Round your answers to two decimal places....
-
The Moon has a period of 27.3 days and a mean distance of 3.9 x 105 km from its center to the center of Earth. Use Kepler's Third Law. (At the end convert days into seconds) a) Find the period of...
-
Summary of intervention orders that ASIC imposes on banning short term lending model to protect consumers from predatory lending. Explain with examples
-
On January 1, year 1, The company borrows $50,000 to purchase a new vehicle by agreeing to a 6.0%, 5-year loan with the bank. Payments are due at the end of each month with the first installment...
-
2. The length of stay in a certain cancer Hospital were organized into a frequency distribution. The mean length of stay was 28 days, the medial 25 days and modal length is 23 days. The standard...
-
A simplified vibration analysis of an airplane considers bounce and pitch motions (Fig. 6.24(a)). For this, a model consisting of a rigid bar (corresponding to the body of the airplane) supported on...
-
Use nodal analysis to determine voltages v1, v2, and v3 in the circuit Fig. 3.76. Figure 3.76 4 S 3i, 2 A 4A
-
Figure 8 shows velocity curves for two cars, A and B, that start side by side and move along the same road. What does the area between the curves represent? Use the Midpoint Rule to estimate it....
-
What product would you expect to obtain from Grignard reaction of an excess of phenyl magnesium bromide with dimethyl carbonate, CH 3 OCO 2 CH 3 ?
-
Treatment of 5-aminopentanoic add with DCC (dicyclohexylcarbodiimide)) yields a lactam. Show the structure of the product and the mechanism of the reaction.
-
Outline methods for the preparation of acetophenone (phenyl methyl ketone) starting from the following: (a) Benzene (b) Bromobenzene (c) Methyl benzoate (d) Benzonitrile (e) Styrene
-
(chi-square) - We will be using the chapek9 data set on jamovi. Test the research hypothesis that, robots prefer data and humans prefer puppies (**note** start with adding the flowing filter: fx =...
-
Vous avez r cemment t embauch par Scheuer Media Inc. pour estimer le co t de ses actions ordinaires. Vous avez obtenu les donn es suivantes : D _ 1 = 1 , 7 5 $ ; P _ 0 = 4 2 , 5 0 $ ; et g = 7 , 0 0...
-
Given the following information for the Duke Tire Company, find ROA (Return on Assets): Debt ratio (D/A) = 0.37 (expressed as a decimal) Total asset turnover ratio (S/A) = 1.16 Sales (S) = $10,000...

Study smarter with the SolutionInn App


