Consider the following samples of gases at the same temperature. Arrange each of these samples in order
Question:
Consider the following samples of gases at the same temperature.
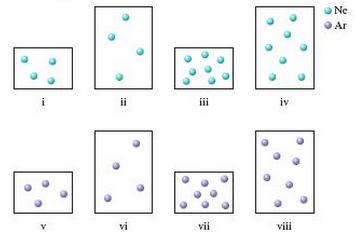
Arrange each of these samples in order from lowest to highest.
a. Pressure
b. Average kinetic energy
c. Density
d. Root mean square velocity
Some samples of gases may have equal values for these attributes. Assume the larger containers have a volume twice the volume of the smaller containers and assume the mass of an argon atom is twice the mass of a neon atom.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





