Draw as many resonance structures as you can for the following species. Adding appropriate formal charger to
Question:
Draw as many resonance structures as you can for the following species. Adding appropriate formal charger to each:
(a) Nitromethane,
(b) Ozone,
(c) Diazomethane,
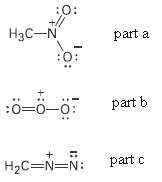
Transcribed Image Text:
:0: +// H3C-N part a :0: part b :0=0-0: part c H2C=N=N: :o:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a c 0 HC...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw as many resonance structures as you can for the followingspecies: (b) :NH2 H2N-C=NH2 (c) (a) :0: H --- () HC $H2 (e) H %H
-
Suggest the best syntheses that you can for the following amines, beginning each with any organic compounds that do not contain nitrogen. (a) Butanamine; (b) N-methylbutanamine; (c)...
-
Draw contributing resonance structures for each of the following species, and rank the structures in order of decreasing contribution to the hybrid: a. b. c. d. e. f. CH3C-CH CHCH3 CH3 0 CH3COCH3 +OH...
-
In a plant producing multiple products, would absorption costing overcost or undercost the more complex products? Why?
-
In 2016, Bianca earned a salary of $164,000 from her employer. Determine the amount of FICA taxes and Medicare taxes withheld from her salary.
-
Elmo Inc.s stock is currently selling at $60 per share. For each of the following situations (ignoring brokerage commissions), calculate the gain or loss that Courtney Schinke realizes if she makes a...
-
Zurich Manufacturing incurs unit costs of \($8.50\) (\($5.50\) variable and \($3\) fixed) in making a subassembly part for its Finished product. A supplier oilers to make 10.000 of the assembly part...
-
Robert Campbell and Carol Morris are senior vice presidents of the Mutual of Chicago Insurance Company. They are codirectors of the companys pension fund management division, with Campbell having...
-
Question 3 Asset Valuation Sandhill Industries Corp. purchased the following assets and also constructed a building. All this was done during the current year. Assets 1 and 2 These assets were...
-
Western Family Steakhouse offers a variety of low-cost meals and quick service. Other than management, the steakhouse operates with two full-time employees who work 8 hours per day. The rest of the...
-
Which of the following pairs represent resonance structures? (a) CH3C=N-O: and CH3C=N-o: (b) :0: :C C0: and (d) (c) : and :CH2-N CH2=N NH3 and NH2
-
Carbocations, ions that contain a trivalent, positively charged carbon atom, react with water to give alcohols. How can you account for the fact that the following carbocation gives a mixture of two...
-
To minimize the potential for gastrointestinal disease outbreaks, all passenger cruise ships arriving at U.S. ports are subject to unannounced sanitation inspections. Ships are rated on a 100-point...
-
1. Recall in the Best Investigator case involving the murder of Tanner and Dobbs, that the skull collected from the coffee table was not that of Tanner but rather a female child. Suppose the...
-
Alpha and Beta Companies can borrow for a five-year term at the following rates: Moody's credit rating Fixed-rate borrowing cost Floating-rate borrowing cost Calculate the quality spread differential...
-
How does strategic alignment across various departments impact organizational effectiveness, and what methods can be employed to ensure a cohesive approach to achieving organizational objectives?
-
Draft a contract according to the facts. The facts are in the following page. will need to conduct extensive independent research to prepare the contract. You may use a template for drafting your...
-
On October 1, 2015, Surround Co. was identified as a potentially responsible party by the Environmental Protection Agency. Surround management along with its counsel have concluded that it is...
-
At the end of the first day of business, Quick Clean Laundry has the-assets and liabilities shown below. The owner, Anh Vu, wants to know the amount of her equity in Quick Clean Laundry. Determine...
-
Prove the formula for (d/dx)(cos-1x) by the same method as for (d/dx)(sin-1x).
-
Using the Redlich-Kwong equation of state, compute and plot (on separate graphs) the pressure of nitrogen as a function of specific volume at the two temperatures: a. 110 K b. 150 K
-
Explain in which solvent these reaction are faster: a) HC Br in CHOH or CH-CHOH I b) CHCHCHCH + OH in CHOH or 50% CHOH 50% HO CHICHICH c) CHCHCH + C=N: in CHCHOH or DMSO
-
Explain whether these reactions follow an SN1 or an SN2 mechanism. CH3 T a) CH3-C-Br + CHCO CH3 b) OMS d) + SH CH3 c) CH CH-C-0 T CH3 DMF CHCH-Br + CHOH + CH CO + CHCHOH CH,COH 1-BUOH CHOH CHCCH EtOH
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Consider how Star Valley, a popular ski resort, could use capital budgeting to decide whether the $8 million Blizzard Park Lodge expansion would be a good investment. Assur conce assur Click the icon...
-
Selected T-accounts of Moore Company are given below for the just completed year: Raw Materials Debit Credit Balance 1/1 28,000 Credits Debits 146,000 Balance 12/31 38,000 Manufacturing Overhead...
-
Your company has total receivables outstanding at 31 December 2020 of $20,000. You think that about 2% of these balances will not be collected and wish to make an appropriate allowance. You have not...

Study smarter with the SolutionInn App


