Ethyl dimethylacetoacetate reacts instantly at room temperature when treated with ethoxide ion to yield two products, ethyl
Question:
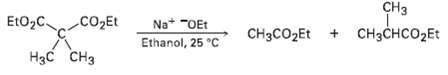
Ethyl dimethylacetoacetate reacts instantly at room temperature when treated with ethoxide ion to yield two products, ethyl acetate and ethyl 2-mcthylpropanoatc. Propose a mechanism for this cleavagereaction.
Transcribed Image Text:
CHз CH3CHCO2ET EtO,C. CO2Et Na* "OEt Ethanol, 25 °C CH3CO2ET + Нзс Cнз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Go CHCCCH32COEtCHCCCHCOEt addition of ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for each reaction. (a) (b) (c) (d) OH H2SO heat OCH + CH3OH H20 CH2OH H2So4 heat CH OCH2CH CH CH,CH2OH (a minor product)
-
Propose a mechanism for each reaction. (a) (b) Ht H3C CH3 OH H SOA Ph Ph Ph Ph
-
Propose a mechanism for the acid-catalyzed condensation of n-propyl alcohol to n-propyl ether, as shown above. When the temperature is allowed to rise too high, propene is formed. Propose a mechanism...
-
Two positive charges, each with charge q = 2.5 nC, are placed as shown in the diagram. The distance d 0.42 m. Find the net electric potential at the point x = 0, y = 0.12 m.
-
Consider an industry in which firms can expect to sell 1000 units annually at a price of P. Before firms enter, they do not know their production costs with certainty. Instead, they believe that unit...
-
In FIGURE EX10.26, what is the maximum speed of a 2.0 g particle that oscillates between x = 2.0 mm and x = 8.0 mm? U (J) 5- 4 3- 2- 0+ x (mm) 4 FIGURE EX10.26
-
Think like an owner and act like a manager.
-
On January 1, 2014, Wolf Creek Country Club purchased a new riding mower for $15,000. The mower is expected to have a 10-year life with a $1,000 salvage value . What journal entry would Wolf Creek...
-
If a bank's return on equity (ROE) is 4.5% and its return on assets (ROA) is 1.5%, what is the bank's assets/equity ratio (equity multiplier)? show your work
-
Consider a fixed partitioning scheme with equal-size partitions of 216 bytes and a total main memory size of 224 bytes. A process table is maintained that includes a pointer to a partition for each...
-
Give the structures of the possible Claisen condensation products from the following reactions. Tell which, if any, you would expect to predominate in each case. (a) CH3CO2Et + CH3CH2CO2Et (b)...
-
In contrast to the rapid reaction shown in Problem 23.40, ethyl acetoacetate requires a temperature over 150 ?C to undergo the same kind of cleavage reaction. How can you explain the difference in...
-
What is the UML, and how is it determined?
-
List the model assumptions for one-way ANOVA and briefly explain how to assess them.
-
Remember that a correctly labeled graph requires that you label all axes, curves, and equilibrium point values. The word "calculate" means you must show your work. Assume the market for Good Z is in...
-
Answer the following questions by writing a paragraph or two in English. (a) [easy] Previously we defined probability as P(A) = Describe a situtation where this fails to produce the correct...
-
Find f''(x). f(x)=5x-14x- 612x f'(x)=
-
Simplify. 32-6 3-6
-
Explain how management implements motivational strategies.
-
The Thomas Corporation was organized on Jan. 1, 2020. On Dec. 31, 2021, the corporation lost most of its inventory in a warehouse fire before the year-end count of inventory was to take place. just...
-
Write the Lewis structure for each molecule or ion. a. H 3 COCH 3 b. CN c. NO 2 d. ClO
-
Give a correct name for each compound. (a) (b) (c) (d) (e) (f) CH CH3CH22C CHCH Cl CH,CI
-
(a) Draw and name all five isomers of formula C3H5F. (b) Draw all 12 acyclic (no rings) isomers of formula C4H7Br. Include stereoisomers. (c) Cholesterol, C27H46O, has only one pi bond. With no...
-
Draw and name all stereoisomers of 3-chlorohepta-2,4-diene (a) Using the cis-trans nomenclature. (b) Using the E-Z nomenclature.
-
Palisade Creek Co. is a merchandising business that uses the perpetual inventory system. The account balances for Palisade Creek Co. as of May 1, 2019 (unless otherwise indicated), are as follows:...
-
1-When accounting for an acquisition, goodwill is the difference between what two things? 2- What factors should be considered when deciding whether an acquisition should be financed with cash or...
-
What is the main friction Fluidity aims to address? REAL STATE

Study smarter with the SolutionInn App


