Fill in the missing reagents a?h in the following scheme: CO2Et CH3 CO2Et 2. f 1. c
Question:
Fill in the missing reagents a?h in the following scheme:
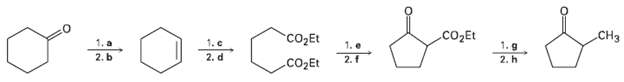
Transcribed Image Text:
CO2Et CH3 CO2Et 2. f 1. c 2. d 1. a 2. b 1. g 1. e 2. h CO2Et
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
Ot 1 a 2 b 10 2 f COEt 19 2h 1 ...View the full answer

Answered By

Wonder Dzidzormenu
As a professional accountant and a teacher, I explain account ing concepts in a more practical way that makes students more connected to the subject.
With over 10 years of teaching accounting , I offer a well constructed , easily understood and in-depth explanations to students questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Fill in the missing reagents below. Me OEt ?| Br Me SH OEt OMe OEt CN
-
Fill in the missing reagents a?c in the following scheme: NH2 CHCH3 CH=CH2 CCH3 b, c CHCH2NCH3 CH CH-CH2
-
Fill in the missing reagents a?d in the following synthesis of racemic methamphetamine from benzene. b, c NHCH3 (R,S)-Methamphetamine
-
The Bay City Parks and Recreation Department is considering building several new facilities, including a gym, an athletic field, a tennis pavilion, and a pool. It will base its decision on which...
-
Provide an example of a firm that has co-specialized assets. Has the firm prospered from them? Why or why not?
-
Suppose you are the sole shareholder of a bank with deposits of $1,200,000 and assets of $1,000,000. There is no reserve requirement. Your liability in the bank is limited by law to your investment...
-
What deal points are most important to the prospective lender?
-
Review the data provided in Exercise 9-1. Metro Industries is considering the purchase of new equipment costing $1,200,000 to replace existing equipment that will be sold for $180,000. The new...
-
\$121,200. The investinent center profit margin is 2205. 545 4538 18368 12.0k
-
Perform an analysis of the sales data for the Vintage Restaurant. Prepare a report for Karen that summarizes your findings, forecasts, and recommendations. Include the following: 1. A time series...
-
The following reactions are unlikely to provide the indicated product in high yield. What is wrong witheach? . , (a) Na* "OEt CHH2CH CHCH CHCH Ethanol (b) .oon CH2CH2CCH3 Na* "OEt + H3 Ethanol "H...
-
How would you prepare the following compounds fromCyclohexanone? la) (b) o C6H5CH, CHC&H5 CH2CH2CN (c) (d) CH2CH=CH2 CO2Et
-
What is the principal difference in job order costing between service and manufacturing firms? Discuss.
-
solve for x 4 . 0 a 2 = 2 . 0 a x
-
BUSINESS SOLUTIONS Comparative Balance Sheets March 3 1 , 2 0 2 2 December 3 1 , 2 0 2 1 Assets Cash $ 8 4 , 7 8 7 $ 5 7 , 8 7 2 Accounts receivable 2 4 , 2 6 7 5 , 0 6 8 Inventory 6 1 4 0 Computer...
-
Solve:z-18=-103.
-
Complete the social penetration exercise and post your reactions in the discussion. PIRATION Purpose: 1. To help you understand the breadth and depth of self-disclosure. 2. To help you see the...
-
The implicit equation of x = sin ( t ) and y = 2 cos ( t ) is:
-
Evaluate the importance of adopting your own leadership style.
-
Information graphics, also called infographics, are wildly popular, especially in online environments. Why do you think infographics continue to receive so much attention? How could infographics be...
-
Determine if a bond between each pair of atoms would be pure covalent, polar covalent, or ionic. a. C and N b. N and S c. K and F d. N and N
-
E1 eliminations of alkyl halides are rarely useful for synthetic purposes because they give mixtures of substitution and elimination products. Explain why the sulfuric acid-catalyzed dehydration of...
-
The following reaction is called the pinacol rearrangement. The reaction begins with an acid-promoted ionization to give a carbocation. This carbocation undergoes a methyl shift to give a more...
-
Propose a mechanism to explain the formation of two products in the following reaction. CH2Br NBS, hv Br
-
The company sold merchandise to a customer on March 31, 2020, for $100,000. The customer paid with a promissory note that has a term of 18 months and an annual interest rate of 9%. The companys...
-
imer 2 0 2 4 Question 8 , PF 8 - 3 5 A ( similar to ) HW Score: 0 % , 0 of 1 0 0 points lework CH 8 Part 1 of 6 Points: 0 of 1 5 Save The comparative financial statements of Highland Cosmetic Supply...
-
An investor wants to purchase a zero coupon bond from Timberlake Industries today. The bond will mature in exactly 5.00 years with a redemption value of $1,000. The investor wants a 12.00% annual...

Study smarter with the SolutionInn App


