Indicate whether these species are weaker or stronger bases than hydroxide ion. The Ka is or pKa
Question:
Indicate whether these species are weaker or stronger bases than hydroxide ion. The Ka is or pKa values are for the conjugate acids.
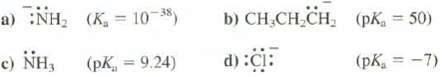
Transcribed Image Text:
a) :NH, (K = 10-38) c) NH, (PK, = 9.24) b) CH₂CH₂CH₂ d) :CI: (pK₁ = 50) (pK₁ = -7)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
The weaker the acid the stronger its conjugate base if the ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Indicate whether these compounds are weaker or stronger acids than water (the K a for water is 1.8 x 10 ?16 ; the p K a is 15.74): (K=100) a) HCIO4 c) HOCOH (PK = 6.35) b) HCCH (pK = 25) d) CH3CH...
-
The pKa of the anilinium ion (C6H5NH3) is 4.63. On the basis of this fact, decide whether aniline (C6H5NH2) is a stronger or weaker base than methylamine.
-
The pKa values of oxaloacetic acid are 2.22 and 3.98. a. Which carboxyl group is more acidic? b. The amount of hydrate present in an aqueous solution of oxaloacetic acid depends on the pH of the...
-
1. If your job were to allocated one large amount of money to combat the illegal drug problem in America, (assume strong addictive characteristics to these drugs). Would you attack the supply or...
-
In your own words, explain two of the sources of inaccuracies in an ABB system?
-
The first audit of the books of Gomez Limited was recently carried out for the year ended December 31, 2017. Gomez follows IFRS. In examining the books, the auditor found that certain items had been...
-
E 5-4 Upstream sales Pop Corporation owns an 80 percent interest in Son Corporation and at December 31, 2016, Pops investment in Son on an equity basis was equal to 80 percent of Sons stockholders...
-
Green Thumb operates a commercial plant nursery where it propagates plants for garden centers throughout the region. Green Thumb has $ 4,800,000 in as-sets. Its yearly fixed costs are $ 600,000, and...
-
Hello, Please help me complete this tax return. ... 7. Taxable income: $50613 Refund:$9486 Please complete the 2019 federal income tax return for Holly Hansen. Ignore the requirement to attach the...
-
During a period of severe inflation, a bond offered a nominal HPR of 80% per year. The inflation rate was 70% per year. a. What was the real HPR on the bond over the year? b. Compare this real HPR to...
-
Provide the values for the missing K a or p K a in the following examples: (a) p K a = 4; K a =? (b) K a = 1 x 10 16 ; p p K a =? (c) p K a = 38; K a =? (d) K a = 1 x 10 6 ; p K a =?
-
Using the information available in Figure 4.2, predict the position of the equilibrium in these reactions; that is, predict whether there is a higher concentration of reactants or products present at...
-
Repeat the analysis shown in Example 5.3 for finding the location of a decision boundary using the following information: (a) The prior probabilities are P(Crocodile) = 2 P(Alligator). (b) The prior...
-
The relationship between income and savings, let's look back to the recent credit crisis that sent our economy into the greatest financial crisis since the Great Depression. Watch this short video...
-
Jos Lpez has $15,000 in a 6-year certificate of deposit (CD) that pays a guaranteed annual rate of 4%. Create a timeline showing when the cash flows will occur. (6 points) 2. Oliver Lpez deposits...
-
PROBLEM SET #2 At a large urban college, about half of the students live off campus in various arrangements, and the other half live on campus. Is academic performance dependent on living...
-
Post a compelling argument stating whether leaders are born, made, or a combination of both. Drawing from the discussion of the two current peer-reviewed articles you identified, support your...
-
Unicorn Inc. builds commercial jets and calculate the cost for each jet. For each item below, indicate whether it would be most likely classified as direct labor (DL); direct materials (DM);...
-
What are some of the specific crisis situations that you might anticipate as a hospitality manager?
-
To balance the chemical equation SiH3 + O2 SiO2 + HO, you could introduce coefficients a, b, c, d and write aSiH3 + bO2 cSiO + dHO then write linear equations for each element. The equation for Si...
-
Find the sum of the series where |x| 00 .". n=0
-
Identify the following reactions as additions, eliminations, substitutions, orrearrangements: (a) CH3CH2Br + NACN CH3CH2CN (+ NaBr) (b) Acid (+ H20) OH cataiyst (c) Heat NO2 "O. (d) Light (+ HNO2)...
-
What is the difference between a transition state and an intermediate?
-
Draw an energy diagram for a one-step reaction with Keq < 1. Label the parts of the diagram corresponding to reactants, products, transition state, G, and G++. Is G positive or negative?
-
A company is evaluating a new 4-year project. The equipment necessary for the project will cost $3,300,000 and can be sold for $650,000 at the end of the project. The asset is in the 5-year MACRS...
-
You have just been hired as a new management trainee by Earrings Unlimited, a distributor of earrings to various retail outlets located in shopping malls across the country. In the past, the company...
-
I need to see where the calculations for this problem come from plz. 5. Award: 4.00 points Lucido Products markets two computer games: Claimjumper and Makeover. A contribution format income statement...

Study smarter with the SolutionInn App


