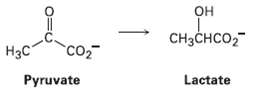
Lactic acid buildup in tired muscles results from reduction of pyruvate. If the reaction occurs from the
Question:
Lactic acid buildup in tired muscles results from reduction of pyruvate. If the reaction occurs from the Re face, what is the stereochemistry of theproduct?
Transcribed Image Text:
OH он CH3CHCO2 H3C CO2 Lactate Pyruvate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Strategy Use the strategy in the previous problem to id...View the full answer

Answered By

David Muchemi
I am a professional academic writer with considerable experience in writing business and economic related papers. I have been writing for my clients who reach out to me personally after being recommended to me by satisfied clients.
I have the English language prowess, no grammatical and spelling errors can be found in my work. I double-check for such mistakes before submitting my papers.
I deliver finished work within the stipulated time and without fail. I am a good researcher on any topic especially those perceived to be tough.
I am ready to work on your papers and ensure you receive the highest quality you are looking for. Please hire me to offer my readily available quality service.
Best regards,
4.60+
27+ Reviews
61+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
If lactic acid is the buildup product of strenuous muscle activity, why is sodium lactate often given to hospital patients intra-venously?
-
The pyruvate ( lactate reaction in animals is reversible, but the pyruvate ( ethanol fermentation in yeast is irreversible. Explain.
-
What results from applying Gauss-Jordan reduction to a nonsingular matrix?
-
PIM Industries, Inc., manufactures electronics components. Each unit costs $30 before the final test. The final test rejects, on average, 5 percent of the 50,000 units manufactured per year. The...
-
Rank the magnitudes of these units of thermal energy from greatest to least: a. 1 calorie b. 1 Calorie c. 1 joule
-
Consider the eight investments listed in problem P7-9. Data from P7-9. Consider the following eight investments. a. Investment in 500 shares of Bank of Montreal: Management believes the shares are...
-
Use the PewWorkPlay dataset to assess the relative effects of the following variables on the Use Index: Streaming and Sports index: XYracewb, XYsex, and age. Create a professional-looking table that...
-
Thomas Industries has the following patents on its December 31, 2014, balance sheet. The following events occurred during the year ended December 31, 2015. 1. Research and development costs of...
-
XY Inc. provides the following data for June 2019 when 13,000 Units are manufactured: Standard Material Cost (Per Unit) 6.20 kg @ $ 10.25/kg Actual Material Cost (Per Unit) 6.85 kg @ $ 13.8/kg...
-
Verify that Stokes Theorem is true for the given vector field F and surface S. F(x, y, z) = -2yz i + y j + 3x k, S is the part of the paraboloid z = 5 - x 2 - y 2 that lies above the plane z = 1,...
-
Identify the indicated faces of carbon atoms in the following molecules as Re orSi: (b) (a) H-2 "CH2 - H Hydroxyacetone Crotyl alcohol
-
The aconitase-catalyzed addition of water to cis-aconitate in the citric acid cycle occurs with the following stereochemistry. Does the addition of the OH group occur on the Re or the Si face of the...
-
Bird-Bath, inc., experienced four situation for its supplies. Compute the amounts that have been left blank for each situation For situations 1 and 2, Journalize the needed transaction. Consider each...
-
2 4. A rod of length 2 cm makes an angle rad with the principal axis of a thin convex lens. The lens has a focal 3 40 3 cm from the object as shown in the figure. The height of the length of 10 cm...
-
7. Consider an LC circuit, with inductance L = 0.1 H and capacitance C = 103 F, kept on a plane. The area of the circuit is 1 m. It is placed in a constant magnetic field of strength Bo which is...
-
Beach 10. The figure shows a circuit having eight resistances of 10 each, labelled R1 to R8, and two ideal batteries with voltages & = 12 V and 2 = 6 V. 1 R B1 Rs R R Which of the following...
-
12. Three plane mirrors form an equilateral triangle with each side of length L. There is a small hole at a distance /> O from one of the corners as shown in the figure. A ray of light is passed...
-
15. A small circular loop of area A and resistance R is fixed on a horizontal xy-plane with the center of the loop always on the axis of a long solenoid. The solenoid has m turns per unit length and...
-
Describe the bar setup. LO.1
-
Create a data model for one of the processes in the end-of-chapter Exercises for Chapter 4. Explain how you would balance the data model and process model.
-
Summarize each of the simple gas laws (Boyles law, Charless law, and Avogadros law). For each, explain the relationship between the two variables and also state which variables must be kept constant.
-
Draw all enol isomers of the following compounds. If there are none, explain why. (a) 2-methylpentanoic acid (b) Benzaldehyde
-
Draw all enol isomers of the following compounds. If there are none, explain why. (a) 2-methylpentanoic acid (b) Benzaldehyde
-
Crossed aldol condensations can be carried out if one of the carbonyl compounds is unusually acidic. Explain why the aldol condensation of acetone with itself does not compete with the crossed aldol...
-
Estimate the intrinsic value of the stock company ABC. Dividends were just paid at $8 per share and are expected to grow by 5%. You require 20% on this stock given its volatile characteristics. If...
-
Crane, Inc., a resort management company, is refurbishing one of its hotels at a cost of $6,794,207. Management expects that this will lead to additional cash flows of $1,560,000 for the next six...
-
Match each of the following transactions with the applicable internal control principle that is being violated

Study smarter with the SolutionInn App


