The aconitase-catalyzed addition of water to cis-aconitate in the citric acid cycle occurs with the following stereochemistry.
Question:
The aconitase-catalyzed addition of water to cis-aconitate in the citric acid cycle occurs with the following stereochemistry. Does the addition of the OH group occur on the Re or the Si face of the substrate? What about the addition of the H? Does the reaction have syn or antistereochemistry?
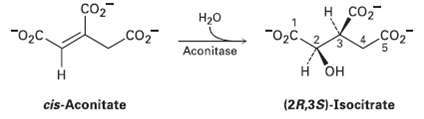
Transcribed Image Text:
н со Но "02c. "O2C. co2 3. Aconitase H OH он (2R,3S)-Isocitrate cis-Aconitate 3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Addition of OH takes place on the Re face of aconi...View the full answer

Answered By

Ishrat Khan
Previously, I have worked as an accounting scholar at acemyhomework, and have been tutoring busines students in various subjects, mostly accounting. More specifically I'm very knowledgeable in accounting subjects for college and university level. I have done master in commerce specialising in accounting and finance as well as other business subjects.
5.00+
137+ Reviews
431+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The first step in the citric acid cycle is reaction of oxaloacetate with acetyl CoA to give citrate. Propose a mechanism, using acid or base catalysis asneeded. "O2C. 02C H .co2 CO SCOA Citrate...
-
Which of the substances in the citric acid cycle are tricarhoxylic acids, thus giving the cycle its alternative name?
-
Isocitric acid, an intermediate in the citric acid cycle of food metabolism, has the systematic name (2R, 3S)-3-carboxy-2-hydroxypentanedioic acid. Draw the Structure.
-
Foreman Publishing Companys income for the most recent quarter was $500,000, and the average net book value of assets during the quarter was $1.5 million. If the company has a required rate of return...
-
Consider a bar 1 m long that expands 0.6 cm when heated. Show that when similarly heated, a 100-m bar of the same material becomes 100.6 m long.
-
9 . 5 - 4 . Ledolter and Hogg (see References) note that percent yields from a certain chemical reaction for changing temperature (factor A), reaction time (factor B), and concentration (factor C)...
-
The equity method is often described as a one-line consolidation. Discuss the similarities of the equity method and consolidated reporting. Discuss how these methods differ in their financial...
-
Multiple product break-even analysis Eaton Company manufactures two products. The budgeted per-unit contribution margin for each product follows: Eaton expects to incur annual fixed costs of...
-
Diaz Enterprises makes 12 oz, 16 oz, and 20 oz coffee mugs. The company is considering dropping the 16 oz. size. Financial information on the 16 oz product line is shown below. Sales: $22,000...
-
If you want to start a new business, what form of ownership will you choose and why?
-
Lactic acid buildup in tired muscles results from reduction of pyruvate. If the reaction occurs from the Re face, what is the stereochemistry of theproduct? OH CH3CHCO2 H3C CO2 Lactate Pyruvate
-
Which of the following structures are identical? (Yellow-green =Cl) (a) (b) (c) (d)
-
An airplane flies at Mach 0.8 at an altitude of \(10 \mathrm{~km}\). Its engine draws in air from the free stream and raises its pressure by a factor of 80 by the time it exits the engine's...
-
Suppose the real risk - free rate is 3 . 5 0 % , the average future inflation rate is 2 . 5 0 % , a maturity premium of 0 . 2 0 % per year to maturity applies, i . e . , MRP = 0 . 2 0 % ( t ) , where...
-
An electricity company pays a dividend $ 1 . 6 4 per share and the share price is $ 2 7 . a ) If the investors believe that the dividend growth rate will be 3 % annually, what will be the yield that...
-
Tektron Industries applies overhead on the basis of 200% of direct labor cost. Job No. 275 is charged with $30,000 of direct materials costs and $40,000 of manufacturing overhead. What are the total...
-
A polymer is injected into a mold at 205 C. The mold is 35 C. The part is 6 mm thick, and can be modeled as a plate. The glass transition temperature of the polymer is 125 C. The density of the...
-
x 2-1.02 X T(X. y) Tot X=X1 xupuRy & incident angle a: reflection angle 090-8 m = to 8 riy-y = m (x-x) ton & = x1 tonB=26, tonx = 26 X1 what is the ton y and r.
-
Use the Laplace transform method to determine the response of an undamped SDOF system initially at rest in equilibrium when subject to a symmetric triangular pulse of magnitude \(F_{0}\) and total...
-
Create a data model for one of the processes in the end-of-chapter Exercises for Chapter 4. Explain how you would balance the data model and process model.
-
A 1.25-g gas sample occupies 663 mL at 25 C and 1.00 atm. What is the molar mass of the gas? a) 0.258 g/mol b) 0.0461 g/mol c) 3.87 g/mol d) 46.1 g/mol
-
Explain the following findings. (a) One full equivalent of base must be used in the Claisen or Dieckmann condensation. (b) Ethyl acetate readily undergoes a Claisen condensation in the presence of...
-
A student, Cringe Labrack, has suggested each of the faulty synthetic procedures shown in Fig. P22.73. Explain why each one cannot be expected to work. NadEt CHAI NaOEt PhBt a) CH,CH CO,Et (c OH CH...
-
A student, Cringe Labrack, has suggested each of the faulty synthetic procedures shown in Fig. P22.73. Explain why each one cannot be expected to work. NadEt CHAI NaOEt PhBt a) CH,CH CO,Et (c OH CH...
-
Consider two events related to customer satisfaction at a restaurant: Let F denote the event that a customer rates the quality of food as excellent. The probability of event F is 0.75. Let S denote...
-
The maximum deflections of the bridge girder were measured under 10 different truckloads when the loads ranged from 100 kN to 300 kN. The load, (kN), and the associated maximum deflection, (mm), are...
-
Find the maturity value, discount period, discount, and proceeds for a promissory note that is discounted at 1 6 1 6 % . Use banker's interest of 3 6 0 days.

Study smarter with the SolutionInn App


