Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
Question:
Predict the products of the following reactions:
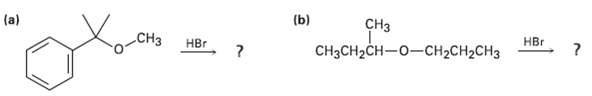
Transcribed Image Text:
(a) (b) CHз CH3CH2CH-0-CH2CH2CH3 CHз НЕr HBr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a First notice the substitution pattern of the ether Bo...View the full answer

Answered By

Joram mutua
I am that writer who gives his best for my student/client. Anything i do, i give my best. I have tutored for the last five years and non of my student has ever failed, they all come back thanking me for the best grades. I have a degree in economics, but i have written academic papers for various disciplines due to top-notch research Skills.In additional, I am a professional copywriter and proofreader.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant. H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e)...
-
Predict the products of the following reactions: CH (b) CH (a) 1. (NH2)2C=S 2. NaOH, H20 -CH CH2CH2CH2Br Hr SCH2CH3 (d) (c) Br2, ? H0z. 2 SH
-
Predict the products of the following reactions. When more than one product is expected, predict which will be the major product. (a) (b) (c) (d) OH H2SO4 heat H3PO4 heat BrNaocH, CH CH H,SO4 heat OH
-
Which is found in the java.sql package that come with the standard JDK? A. Only DerbyDriver B. Only MySqlDriver C. Only OracleDriver D. DerbyDriver, MySqlDriver, OracleDriver E. Only DerbyDriver and...
-
What are some of the issues with using financial metrics to evaluate IT?
-
Geneticists hypothesized that one reason for a prolonged lifespan observed in yeast cells treated with the common drug ibuprofen stems from inhibiting the uptake of certain amino acids. In an...
-
Brooks Co. purchases various investments in trading securities at a cost of $66,000 on December 27, 2008. (This is its first and only purchase of such securities.) At December 31, 2008, these...
-
The following balance sheet and income statement data were taken from the records of L. L. Beeno for the year ended December 31, 2015: Prepare the operating section of the statement of cash flows,...
-
If total equities on August 1 were $70,381, what were total equities on August 31? X Company, a merchandiser, had the following transactions in August: 1. Borrowed $22,000 from a bank. 2. Bought...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
Rank the following halides in order of their reactivity in the Williamson synthesis: (a) Bromo ethane, 2-bromopropane, bromo benzene (b) Chloro ethane, bromo ethane, 1-iodopropene
-
Write the mechanism of the acid-catalyzed cleavage of tert-butyl cyclohexyl ether to yield Cyclohexanol and 2-methylpropene.
-
Describe the role of sustainable human capital in multinational firms. LO2
-
Given forecast errors of 4, 8, and -3, what is the MAD? What is the MSE?
-
Padgett Rentals can purchase a van that costs \($48,000\) ; it has an expected useful life of three years and no salvage value. Padgett uses straight-line depreciation. Expected revenue is...
-
Rainwater Corp. expects to sell 600 umbrellas in May and 400 in June. Each umbrella sells for \($15\). Rainwaters beginning and ending finished goods inventories for May are 75 and 50 units,...
-
Don Moon is the owner of ABC Cleaning. At the beginning of the year, Moon had \(\$ 2,400\) in inventory. During the year, Moon purchased inventory that cost \(\$ 13,000\). At the end of the year,...
-
Agua Ole is a distributor of bottled water. For each of items a through c, compute the amount of cash receipts or payments Agua Ol will budget for September. The solution to one item may depend on...
-
Define countermeasures, network intrusion detection system (NIDS), and data loss prevention system (DLP). List two ways the Target data breach could have been prevented. Explain why theres nothing...
-
During the month, services performed for customers on account amounted to $7,500 and collections from customers in payment of their accounts totaled $6,000. At the end of the month, the Accounts...
-
Redo Problem 4.11 using Aspen Plus. Problem 4.11 Steam is produced at 70 bar and some unknown temperature. A small amount of steam is bled off just before entering a turbine and goes through an...
-
Suggest a possible structure for a compound with the formula C7H12O that has the following IR spectrum and explain yourreasoning: 80 40 20 1718 cm 0- 500 1000 2000 1500 2500 3000 3500 4000 Wavenumber...
-
Suggest a possible structure for a compound with the formula C9H10O that has the following IR spectrum and explain yourreasoning: 100 - 50 1706 em- 4000 3500 3000 2500 2000 1000 500 1500 Wavenamber...
-
Forensic laboratories often have to identify various illicit drug samples. Explain how IR spectroscopy could be used to help distinguish between morphine and heroin. CH3CO, . NH NH CH,CO" ...
-
Transcribed image text
-
QUESTION 20 Assume a company reported the following results Sales Net operating income Average operating assets Margin Turnover Return on investment (ROI) 5300,000 2 $240.000 40% ? 2 What is the net...
-
2. Using the graph provided below, determine the fixed cost, the total variable cost, the variable cost per unit, and the TOTAL COST to produce 60 units. Fixed Cost ______________ Variable Cost...

Study smarter with the SolutionInn App


