Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions.
Question:
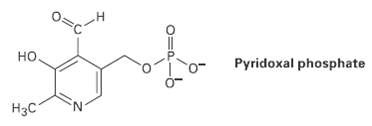
Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions. Tell the hydribization, and predict the bond angles for each non-terminalatom.
Transcribed Image Text:
но. Pyridoxal phosphate Нас 0=
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
sp3 sp2 H3C H sp2 sp2 sp2 sp2 5p3 Pyridoxa...View the full answer

Answered By

Pharashram rai
i am highly creative, resourceful and dedicated freelancer an excellent record of successful classroom presentations and writing . I have more than 4 years experience in tutoring students especially by using my note making strategies and engineering field . Especially adept at teaching methods of maths and writing , and flexible teaching style with the willingness to work beyond the call of duty. Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with .
4.80+
65+ Reviews
270+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the molecular structure and the bond angles for each molecule or ion in Exercises 57, 58, and 60.
-
Predict the molecular structure and the bond angles for each of the following. a. SeO3 b. SeO2 c. PCl3 d. SCl2 e. SiF4
-
Predict the molecular structure and the bond angles for each of the following. (See Exercises 89 and 90.) a. XeCl2 b. ICl3 c. TeF4 d. PCl5
-
In the Akerlof example, the individuals are treated as indifferent to risk. What would you expect to see in these markets if individuals wanted to avoid risk? What if there were some risk lovers?
-
Glen and Diane Okumura (both age 48) are married, file a joint return, and live at 39 Kaloa Street, Honolulu, HI 96815. Glen's Social Security number is 111-11-1111, and Diane's is 123-45-6789. The...
-
Firms should formulate and implement strategies from an environmental perspective. List eight ways firms can do this.
-
Lauren Inc. makes three products that can be sold at split-off or processed further and then sold. The joint cost for April is \(\$ 1,080,000\). The number of ounces in a bottle of each product is:...
-
Hardware Manufacturing Company Limited, a closely held corporation, has operated since 1994 but has not had its financial statements audited. The company now plans to issue additional share capital...
-
An oil company paid a landowner $30,000 for the mineral rights underlying his property. The well was drilled and equipped at a cost of $900,000. It is estimated that 300,000 barrels of oil will be...
-
Read the following cases. For each, state whether the action or situation shows a violation of the AICPA Code of Professional Conduct; if so, explain why and cite the relevant rule or interpretation....
-
What kind of hydribization do you expect for each carbon atom in the followingmolecules? CH- (b) H. C-H (a) 1 "CH-CH CH2 C=C H2N H. Procaine Vitamin C (ascorbic acid)
-
Why do you suppose no one has ever been able to make cyclopentyne as a stable molecule? Cyclopentyne
-
Calculate y'. y = ln sec x
-
The annual Cost of Goods Sold for a company is expected to be 82,000. The beginning inventory balance is 25,000. The ending inventory balance is expected to be 21,000. All purchases are on credit....
-
Listed below are specific audit procedures. For each procedure indicate the type of error the procedure would be likely to detect and the broad category of assertion to which it is related. a....
-
Substantive tests include (1) tests of the details of transactions, (2) tests of the details of balances, and (3) analytical procedures. Listed below are several specific audit procedures. Identify...
-
The Fancy Umbrella Company makes recyclable beach umbrellas. The production process requires 3 square meters of plastic sheeting and a metal pole. The plastic sheeting costs \($0.50\) per square...
-
Day, a CPA who has been practicing alone, decides to "sell his practice" to Knight. As a part of the transaction Knight asks Day to turn over to her all his files and work papers. One client, John...
-
What is Hookes Law ? Derive an expression for relationship between elastic constants.
-
Drainee purchases direct materials each month. Its payment history shows that 65% is paid in the month of purchase with the remaining balance paid the month after purchase. Prepare a cash payment...
-
Can a fluid obeying the virial equation of state have a vapor-liquid transition?
-
Show energy level diagrams for the MOS of these compounds: a) H-C=C-CH3 b) CH0H c) CH3-C-H
-
The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule. C=C 5 o* * Ethene 5 o Energy
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
Assume that you start with a balance of $3700 on your credit card. During the first month, you charge $600, and during the second month, you charge $750. Assume that your credit card charges a 27%...
-
Metallica Bearings, Incorporated, is a young start-up company. No dividends will be paid on the stock over the next nine years because the firm needs to plow back its earnings to fuel growth. The...
-
Problem 13-2 (Algo) A metal fabricator produces connecting rods with an outer diameter that has a 1 0.01 inch specification. A machine operator takes several sample measurements over time and...

Study smarter with the SolutionInn App


