What kind of hydribization do you expect for each carbon atom in the followingmolecules? CH- (b) H.
Question:
What kind of hydribization do you expect for each carbon atom in the followingmolecules?
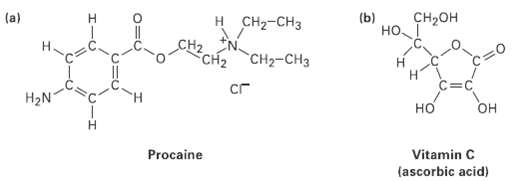
CH-он (b) H. н Cна-сHз (a) но 1 "CH-CHз CH2 C=C сг H2N H. но он Procaine Vitamin C (ascorbic acid)
Step by Step Answer:
a HN St H 302 H sp3 sp CC2 H ...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Organic Chemistry questions
-
What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CH CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHC
-
What dividends do you expect for Goodman Industries stock over the next 3 years if you expect the dividend to grow at the rate of 5% per year for the next 3 years? In other words, calculate D1, D2,...
-
What hybridization do you expect for the atom indicated in red in each of the following species? (a) CH3CO2- (b) PH4+ (c) AlF3 (d) H2C==CH-CH2+
-
Discuss the ways that managed care organizations can infl uence the adoption of new technologies.
-
Review Examples 4 and 6 in the text. In both examples, the taxpayer's AGI is $129,400 even though in Example 6 there is $700 of nonrecaptured 1231 loss from 2015. Explain why the two AGI amounts are...
-
Give an example of consonance other than the one provided by Rumelt in the chapter.
-
Sell or process further Winn Mills processes cotton in a joint process that yields two joint products: fabric and yarn. May's joint cost is \(\$ 120,000\), and the sales values at split-off are \(\$...
-
A Randstad/Harris interactive survey reported that 25% of employees said their company is loyal to them (USA Today, November 11, 2009). Suppose 10 employees are selected randomly and will be...
-
Dove Corporation began its operations on September 1 of the current year. Budgeted sales for the first three months of business are $237,000, $311,000, and $427,000, respectively, for September,...
-
1. Conduct an industry environment analysis. Is Carlsberg A/S in an attractive industry? 2. Identify the reasons for Carlsberg A/S mergers and acquisitions (M&As)? What type of the M&A strategy does...
-
Potassium methoxide, KOCH3, contains both covalent and ionic bonds. Which do you think is which?
-
Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions. Tell the hydribization, and predict the bond angles for each non-terminalatom. . Pyridoxal...
-
Write a program that displays the following table: a 1 2 3 4 aA2 1 16 aA3 1 8 27 64
-
At the end of Year 4, the city owed teachers $60,000 in vacation pay that had not been recorded. The assumption is that these vacations will be taken evenly over the next year. A 60-day period is...
-
In each of the following unrelated circumstances, explain what course of action a CPA should follow in order to discharge his or her professional and ethical responsibilities: 1. Nobles is employed...
-
Humdrum Company is worried about cash flows. The company has \($1,000\) in cash at the start of February. Januarys total sales were \($20,000\) and total sales in February are expected to be...
-
The following questions relate to evidential matter and work papers. Select the best response. a. Audit evidence can come in different forms with different degrees of persuasiveness. Which of the...
-
Indicate which one item in each of the following groups of items of evidence you would consider to be of superior reliability, and briefly explain why. 1 In support of the recorded cost of purchased...
-
Explain belt drive for power transmission.
-
Tell whether the angles or sides are corresponding angles, corresponding sides, or neither. AC and JK
-
Using the Redlich-Kwong equation of state, compute and plot (on separate graphs) the pressure and fugacity of nitrogen as a function of specific volume at the two temperatures a. 110 K b. 150 K
-
Explain why this carbocation is considerably more stable than this structure would suggest: H +C-0-CH, H
-
Explain why one of these anions is much more stable than the other: : a) CH-C-CH-CH b) CH CH3 : CH-C-CH-CH CH-C=N:
-
Explain why one of these carbocations is much more stable than the other: + CH-CH3 CH CH
-
You are considering the purchase of one of two machines required in your production process. Machine A has a life of two years. Machine A costs $ 4 5 initially and then $ 6 5 per year in maintenance....
-
> Question 9 Given the following regarding a share of stock and the overall market: The risk-free rate of return is 1.7% The beta of the stock is 0.72 The expected market premium is 9% What is the...
-
You've collected the following information from your favorite financial website. 52-Week Price Hi Lo Stock (Dividend) Dividend Yield % PE Close Ratio Price Net Change 77.40 10.43 Acevedo .36 2.6 6...

Study smarter with the SolutionInn App


