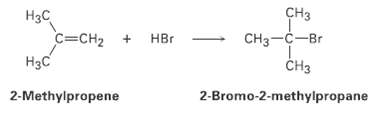
Reaction of HBr with 2-methyipropenc yields 2-hromo-2-methylpropane, what is the structure of the carbocation formed during the
Question:
Reaction of HBr with 2-methyipropenc yields 2-hromo-2-methylpropane, what is the structure of the carbocation formed during the reaction? Show the mechanism of the reaction.
Transcribed Image Text:
CHз CHз—с—Br Нас C=CH2 + HBr Нас сHз 2-Bromo-2-methylpropane 2-Methylpropene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
The mechanism is pictured in Figure The steps 1 Attack of the electrons of th...View the full answer

Answered By

Sumit kumar
Education details:
QUATERNARY Pursuing M.Tech.(2017-2019) in Electronics and Communication Engg. (VLSI DESIGN) from
GNIOT Greater Noida
TERTIARY B.Tech. (2012-2016) in Electronics and Communication Engg. from GLBITM Greater Noida
SECONDARY Senior Secondary School Examination (Class XII) in 2012 from R.S.S.Inter College, Noida
ELEMENTARY Secondary School Examination (Class X) in 2010 from New R.J.C. Public School ,Noida
CERTIFICATION
Summer Training in ‘WIRELESS EMBEDDED SYSTEM’ from ‘XIONEE’ for the six weeks.
EMBEDDED SYSTEM Certificate issued by CETPA INFOTECH for one day workshop.
Certificate of Faculty development program on OPTICAL COMMUNICATION and NETWORKS for one week.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Reaction of HBr with 3-methylcyclohexene yields a mixture of four products: cis- and trans-1-bromo-3-methylcyclohexane and cis- and trans-1-bromo- 2-methylcyclohexane. The analogous reaction of HBr...
-
Reaction of HBr with (R)-3-methyl-3-hexanol leads to racemic 3-brorno-3-mcthylhcxane.Explain. OH CH3CH2CH2CCH2CH3 3-Methyl-3-hexanol H3
-
What is the structure of ATCase?
-
Suppose that in a particular area the consumption of water varies tremendously throughout the year, with average household summer use exceeding winter use by a great deal. What effect would this have...
-
Assume you are 59 years old, want to retire in 6 years, and currently have an investment portfolio valued at $550,000 invested in technology stocks. After talking with friends and relatives, you have...
-
Bowman Company manufactures cooling systems. Bowman produces all the parts necessary for its product except for one electronic component, which is purchased from two local suppliers: Manzer Inc. and...
-
Determine which of the following transactions represent contingent liabilities for June Leasing and indicate the proper accounting treatment at the company's fiscal year-end, by placing the letter of...
-
Elina Siljander owns Elinas Stained Glass in Helsinki, Finland. The business produces and sells three different types of stained glass windows: small, medium, and large. Elina has two full-time...
-
Professor Singh is studying the avoidance behavior of rats to a loud noise in their cage. Professor Singh is most likely a _ Psychologist?
-
A key aspect of H&M's strategy is to provide value to customers by maximizing perceived product benefits, minimizing prices, or both. Given this, how can H&M further increase the value of its...
-
What product would you expect from reaction of cyclohexene with HBr? With HC1 HBr
-
Add curved arrows to the following polar reactions to indicate the flow of electrons in each: :Ci: (a) - :- + -N :CI: -NH (b) H CH: -Br: :Br: (c) :0: :CI: CI "
-
What are the advantages of using subsidiary ledgers?
-
Explain why ABC often reveals that low-volume products are overcosted and high-volume products are under-costed.
-
Ratio analysis always involves which type of arithmetic operation? a. Addition b. Subtraction c. Multiplication d. Division
-
In what ways does ABC product costing differ from traditional product cost methods?
-
Which of the following would not appear on a company's statement of cash flows? a. Cash flow from operating activities b. Net change in cash c. Total assets d. Cash flow for investing activities
-
Which of the following would most likely be classified as a long-term liability? a. Accounts payable b. Notes payable c. Accounts receivable d. Common stock
-
Use one of the sources given in the text (R or http://vassarstats.net/rho.html) to calculate a confidence interval on the correlation between Expenditures for education and SATcombined from Figure...
-
The following selected information was taken from Sun Valley Citys general fund statement of revenues, expenditures, and changes in fund balance for the year ended December 31, 2019: Revenues:...
-
In which of the following is the central atom sp hybridized? (a) BeCl 2 ; (b) BCl 3 ; (c) CCl 4 ; (d) NCl 3 ; (e) None of these.
-
A compound (N) with the molecular formula C9H10O reacts with osmium tetroxide. The 1H NMR spectrum of N is shown in Fig. 14.31 and the IR spectrum of N is shown in Fig. 14.32. Propose a structure for...
-
The IR and 1H NMR spectra for compound X (C8H10) are given in Fig. 14.33. Propose a structure for compound X.
-
The IR and 1H NMR spectra of compound Y (C9H12O) are given in Fig. 14.34. Propose a structure for Y.
-
A $128m investment partnership has a 23.5% gross return in one year and then liquidates. Fees are 1.5-20 and other expenses are 0.68% of assets. $109m of the $128m is LP capital. a. What incentive...
-
GR is an outsourcing company that provides call centre services to a range of clients. As a result of technical advances in telecommunication equipment, the company's existing telephone system is...
-
A company issues a 15-year $6500 bond, redeemable at 99 with bond interest atj4= 6%. The bond is callable at the end of 5 years for $4830 or at the end of 10 years for $6405. Determine the price to...

Study smarter with the SolutionInn App


