Show how this synthesis might beaccomplished: Br CH3 CH3 HO from CN -
Question:
Show how this synthesis might beaccomplished:
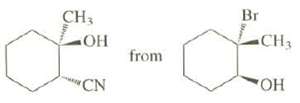
Transcribed Image Text:
Br CH3 CH3 HO from "CN Но-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
Br CH3 O...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show how an acetoacetic ester synthesis might be used to form a -sikeone such as heptane- 2,6-dione.
-
Show how you would use the malonic ester synthesis to make the following compounds. (a) (b) (c) CH,CH C-OH
-
Show how you would use the acetoacetic ester synthesis to make the following compounds. (a) (b) (c) CH-CHC CH CHCH3 CH3
-
Jane Doe is single and claims 2 withholding allowances. If her weekly gross earning is $1,450, then determine the Federal Withholding Tax.
-
Direct and indirect labour: manufacturer Sharpedge Cutlery manufactures kitchen knives. One of the employees. whose job is to cut out wooden knife handles, worked 49 hours during a week in January....
-
Given the following selected account balances of Delray Mfg., prepare its schedule of cost of goods manufactured for the year ended December 31, 2017. Include a listing of the individual overhead...
-
What is the difference between using brainstorming and the Delphi technique for risk identification? What are some of the advantages and disadvantages of each approach? Describe the contents of a...
-
Cholla Companys standard fixed overhead rate is based on budgeted fixed manufacturing over-head of $10,200 and budgeted production of 30,000 units. Actual results for the month of October reveal that...
-
Sheridan Biotech management plans a $100 million IPO in which the offering price to the public will be $80 per share. The company will receive $47.50 per share. The firms legal fees, SEC registration...
-
Fortune magazine's list of the world's most admired companies for 2014 is provided the data contained in the WEB file named Admired Companies (Fortune, March 17, 2014). The data in the column...
-
Show how these compounds could be synthesized from alkylhalides: a) Heptane b) PHCH,CH,CH,NH, c) PHCH,C=CH d) e) HO, OH f) g) H.C
-
What is wrong with these reactions explain. CI + NaOCH3 OCH3 + NaCl a) + HBr Br . b) CH,OH + CH;0 OCH3 CI d)
-
For saving energy, bicycling and walking are far more efficient means of transportation than is travel by automobile. For example, when riding at 10.0 mi/h a cyclist uses food energy at a rate of...
-
A farmer has an acre of specialty vegetables and is preparing for the summer harvest. Historically, this acre has yielded an average of 2,100 lbs of product with a standard deviation of 950 lbs. A...
-
Solve 3x 82+22 = (4).
-
(c) Compute EVPI and EVSI (in thousands of dollars). (Round your answers to one decimal place.) EVPI $ 3.6 EVSI $ 3.6 Xthousand x thousand Discuss whether the firm should consider a consulting expert...
-
Question 9 (1 point) If the common law requires employees of a bar establishment to monitor a potentially intoxicated patron and to possibly make an effort to intervene if there is an indication the...
-
B. A velocity potential is given by the equation: Q = x-y 3. (10 pts) Short answer, What special characteristics of the velocity potential make it very useful in identifying a type of flow and...
-
In what circumstances might you want to create a new variable?
-
The domain of the variable in the expression x 3/x + 4 is________.
-
What is the probability of getting at least 1 tail flipping a coin three times?
-
In light of your answer to Problem 11.61, explain why one of the following isomers undergoes E2 reaction approximately 100 times as fast as the other. Which isomer is more reactive, andwhy? (a) CI...
-
There are eight diastereomers of 1, 2, 3, 4, 5, 6-hexachlorocyclohexane. Draw each in its more stable chair conformation. One isomer loses HCI in an E2 reaction nearly 1000 times more slowly than the...
-
Methyl esters (RCO2CH3) undergo a cleavage reaction to yield carboxylate ions plus iodomethane on heating with LiI in dimethyl form amide: The following evidence has been obtained: (1) the reaction...
-
Comparative financial statements for Weller Corporation, a merchandising company, for the year ending December 31 appear below. The company did not issue any new common stock during the year. A total...
-
Mrquered Mrquered
-
You plan to invest $10,00 today in an investment account earning 5% interest. You then plan to invest an additional $1,000 into this account each year for the next twenty years. How much money will...

Study smarter with the SolutionInn App


