Show how the acetamidomalonate method can be used to prepare the following unusual amino acids from the
Question:
(a)
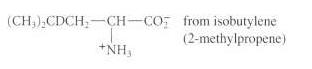
(b)
(c)
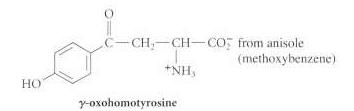
Transcribed Image Text:
(CH) CDCH CH CO from isobutylene (2-methylpropene) +NHs fromn benzaldehyde Ph-CHD-CH-CO; NH C-CH.-CH-CO2 from anisole (methoxybenzene) NH HO -oxohomotyrosine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Every synthesis has the same final steps The challenge in each case ...View the full answer

Answered By

CHARLES AMBILA
I am an experienced tutor with more than 7 years of experience. I have helped thousands of students pursue their academic goals. My primary objective as a tutor is to ensure that students have easy time handling their academic tasks.
5.00+
109+ Reviews
323+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show how you could use the acetamidomalonate method to prepare the following amino acids: (a) Leucine (b) Tryptophan
-
Outline a synthesis for each of the following compounds from the indicated starting material and any other reagents: (a) (b) (c) SC2Hs from pyridine C-O CH CH,CH, from furfural (furan-2-carbaldehyde)...
-
Show how Gabriel syntheses might be used to prepare the following amines. (a) Benzylamine (b) hexan-1-amine (c) g-aminobutyric acid
-
A coil with 190 turns, a radius of 5.0 cm, and a resistance of 12 Ω surrounds a solenoid with 230 turns / cm and a radius of 4.5 cm (Fig. 21-65). The current in the solenoid changes at a...
-
A small manufacturing firm produces two microwave switches, switch A and switch B. Each switch passes through two departments: assembly and testing. Switch A requires 4 hours of assembly and 1 hour...
-
In a double-slit experiment, the fourth-order maximum for a wavelength of 450 nm occurs at an angle of = 90. Thus, it is on the verge of being eliminated from the pattern because cannot exceed 90...
-
The following e-mail with an authentic looking e-mail address and logo was received by the controller of a small business: Required a. What should Justin do about these e-mails? b. What should Big...
-
1. Wayne owns a shaved ice stand. He sells 700 shaved ice cups per month at $1.50 each, making the total revenue $1050. Each shaved ice costs Wayne $0.50 and he has fixed cost of $750, making his...
-
Your firm has expected annual sales of $1.9 million, and has been offered a merchant cash advance of $480,000 with a factor rate of 1.25 and a holdback rate of 20%. a. When do you expect the loan...
-
In an experiment, two identical rocks are simultaneously thrown from the edge of a cliff a distance h0 above the ground. Rock A is thrown vertically upward with speed v0 and rock B is thrown...
-
Complete the reactions given in Fig. P26.61, assuming the amino acid residue is part of a peptide in aqueous solution and is at neither the amino nor the carboxy terminus. (a) (b) plI 8 lysine...
-
When peptides containing a 2, 3-diaminopropanoic acid (DAPA) residue are treated with the Edman reagent and then with acid, a peptide cleavage occurs in addition to degradation of the amino-terminal...
-
A double-pipe, countercurrent heat exchanger is used to cool a hot liquid stream of biodiesel from 60C to 35C. Biodiesel flows on the shell side with a volumetric flow rate of 3 m 3 /h; water is used...
-
Two publishers have approached you with contracts. They are mutually exclusive contracts that pay you upfront. You will have labor costs each year after. $35,000.00 is the pay in the first year of...
-
2) A spool of string of mass M, moment of inertia I about its central axis and radius R falls as it unwinds (the string end is attached to the ceiling). Use energy considerations to find the speed of...
-
Wholesome Foods Inc. (WFI), projects unit sales for its new Organic Trail Mix, as follows: Year Unit Sales 70,000 100,000 3 110,000 4 100,000 65,000 Production of the trail mix will require $5...
-
You bought a bond for $1020 today. The bond's coupon rate is 6.7%, with 12 years left to maturity and a par value of $1,000. What is the yield to maturity of the bond?
-
Mercy, Corp. issued a 8-year bond with a face value of $1,000, a coupon rate of 8.6% and a yield to maturity of 7.9%. What is the current value of the bond?
-
Which of the following are true or false? a. A RNN is designed specifically for processing word sequences. b. A RNN is designed to process any time-series data. c. An RNN has a limit on how far into...
-
Marc Company assembles products from a group of interconnecting parts. The company produces some of the parts and buys some from outside vendors. The vendor for Part X has just increased its price by...
-
The reduction of carbonyl compounds by reaction with hydride reagents (H: ? ) and the Grignard addition by reaction with organo magnesium halides (R: ? + MgBr) are examples of nucleophilic carbonyl...
-
Ethers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Anisole, for instance, results from reaction of sodium phenoxide with iodomethane. What kind of...
-
Identify the functional groups in each of the following molecules: (a) Methionine, an amino acid: (b) Ibuprofen, a pain reliever: (c) Capsaicin, the pungent substance in chilipeppers: ||...
-
Brock and Company would like to purchase equipment for its increased demand for production. The business plans to purchase equipment with a cost of $50,000. The estimated cash inflow for each year is...
-
10 8 00 points eBook Print References Check my work A 30-year maturity, 8% coupon bond paying coupons semiannually is callable in five years at a call price of $1,020. The bond currently sells at a...
-
Ramblin Wreck is a firm specializing in engineering components. The firm is publicly traded and is considering the following project: The project will last 5.00 years with an annual cash flow of...

Study smarter with the SolutionInn App


