Show the product, including stereochemistry, of the following reaction: 1. CH3MgBr, ether 2. H30*
Question:
Show the product, including stereochemistry, of the following reaction:
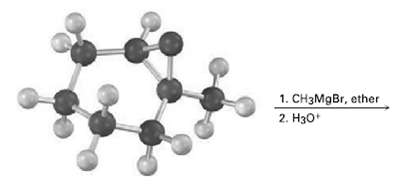
Transcribed Image Text:
1. CH3MgBr, ether 2. H30*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
The Grignard reagent attacks the epoxid...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the product, including stereochemistry that would result from reaction of the following epoxide with HBr.
-
Show how a Wittig reaction can be used to prepare each of the following compounds. In each case, also show how the Wittig reagent would be prepared: (a) (b)
-
Product T is produced for $3.20 per pound, including a $0.20 per pound fixed cost. Product T can be sold without additional processing for $4.10 per pound, or processed further into Product U at an...
-
To load and register a driver, creating a new instance of the Driver class method is better than using the Class.forName() method. (True/False)
-
Describe why continuously learning about technology allows an executive to better analyze threats and opportunities.
-
Use the values in the matrix to solve the equations given. a. P(A U D) = __________ b. P(E U B) = __________ c. P(D U E) = __________ d. P (C U F) =__________ DEF A 58 12 B|10|614 C 825
-
Classifying fixed and variable costs Classify each of the following items of factory overhead as either a fixed or a variable cost. (Include any costs that you consider to be semi variable within the...
-
The narrative and systems flowchart for the Bridgeport LLC cash receipts system are included in Exhibit and Figure, respectively. Using Exhibit and Figure do the following: a. Prepare a control...
-
9 3 9 Ussist Inc. manufactures a number of different products. It's best product, a five - sided rubik's cube, sells for $ 4 8 per unit. The cost to produce the cube is the following: \ table [ [ ,...
-
Why, after 40 years of being self-sufficient and development oriented, did AMP begin aggressively purchasing companies? Was this a good strategic move? What should AMP have done differently?
-
Give IUPAC name for the following compounds (reddish brown =Br): (a) (b) (c)
-
Treatment of the following alkene with a peroxyacid yields an epoxide different from that obtained by reaction with aqueous Br2 followed by base treatment. Propose structures for the two epoxides,...
-
Evaluate the integral. dx e*1-e-2x
-
"The menu looks great, but tell me about the prices one more time," said Nigel, the manager of the Old Dublin Pub. Nigel was talking to Alice Petoskey, the sales representative for Image Custom...
-
Josie is an accounting supervisor at Monk & Sons Realty. She instructs her subordinate, Maria, to make certain accounting entries in the company\'s books that will increase revenue by a material...
-
value based erm approach quantifies individual risk scenarios and enterprise risk exposure in a consistent manner. is this statement correct. Explain
-
Q1. When performing a comparable analysis what considerations are particularly important? Q2. Why do we do both a comparable and a DCF analysis? Don't they give us the same result anyway?
-
To complete this part of the assessment, you willparticipate in a role play scenario to demonstrate your ability to complete skills-based tasks to industry standards.The role play will be observed by...
-
P Ltd acquires 60 per cent of the shares in S Ltd. Balance sheets are then drafted immediately. You are to draw up the consolidated balance sheet. Investment in S Ltd: 1,200 shares Fixed assets Stock...
-
Outline some of the major problems confronting an international advertiser.
-
In normal operation, a paper mill generates excess steam at 20 bar and 400C. It is planned to use this steam as the feed to a turbine to generate electricity for the mill. There are 5000 kg/hr of...
-
Show equations for the major fragmentations you would expect from the molecular ions of these compounds. List the m/z of the productions. CH3 CH2 a) CH;CH-CH,CH-CH; b) CH;CH,CHCH2CH3
-
(a) The base ion in the mass spectrum of 3-ethyl-2-methylpentane occurs at m/z 43m; show the fragmentation that produces this ion. (b) What other fragment would you predict to provide a major peak in...
-
Show equations to account for the major fragment ions that occur at the indicated m/z for thesecompounds: CH,CI b) CH3CH2CH,CH2OH nal: 31 miz 91 c) CH;CH,CCH,CH,CH,CH3 mtz 85, 72, 57
-
Your answer is incorrect. Sheridan Oil Company is considering investing in a new oil well. It is expected that the oil well will increase annual revenues by $ 1 2 2 , 6 2 5 and will increase annual...
-
Bearings & Brakes Corporation ( B&B ) was incorporated as a private company. The company's accounts included the following at June 3 0 : During the month of July, the company had the following...
-
Henry was hired as the Chief Operating Officer (COO) of Zeta Corporation, a publicly-traded company in the technology sector, on April 1, 2022. For the fiscal year 2023, Zeta Corporation compensated...

Study smarter with the SolutionInn App


