On page 351 we showed that the oxidation of glucose and palmitic acid yields 15.94 kJ/g and
Question:
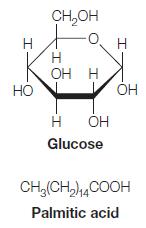
On page 351 we showed that the oxidation of glucose and palmitic acid yields 15.94 kJ/g and 38.90 kJ/g, respectively. Calculate these values in terms of kJ/mol and kJ per carbon atom oxidized for both glucose and palmitic acid.
Transcribed Image Text:
I HO CH₂OH Н OH H -Т Н Glucose ОН I OH CH₂(CH₂) 14COOH Palmitic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
To calculate the energy yield in terms of kJmol and kJ per carbon atom oxidized for both glucose and ...View the full answer

Answered By

Munir Ahmed Jakhro
I am professional Tutor of of Business Courses, I did my four years Bachelor Degree from one of the Top Business schools of World "Institute of Business Administration" in year 2013. Since then I have been working as Tutor of Accounting, Finance tutor on different online platforms like this website. I am have experience of 6 years teaching business courses to students online and offline my professional job at national savings also helped me in accounting understanding .
4.90+
8+ Reviews
10+ Question Solved
Related Book For 

Biochemistry Concepts And Connections
ISBN: 9780134641621
2nd Edition
Authors: Dean Appling, Spencer Anthony-Cahill, Christopher Mathews
Question Posted:
Students also viewed these Sciences questions
-
On page 345 we showed that the oxidation of glucose and palmitic acid yields 15.64 kJ / g and 38.90 kJ / g, respectively. Calculate these values in terms of kJ / mol and kJ per carbon atom oxidized...
-
The oxidation of glucose provides the principal source of energy for animal cells. Assume the reactants are glucose [C6H12O6(s)] and oxygen [O2(g)]. The products are CO2(g) and H2O(l). (a) Write a...
-
The oxidation of glucose (C6H12O6) in body tissue produces CO2 and H2O. In contrast, anaerobic decomposition, which occurs during fermentation, produces ethanol (C2H5OH) and CO2. (a) Using data given...
-
Does the EOQ increase or decrease if estimates of setup (order) costs include fixed, semi-variable, and pure variable costs while inventory-holding costs includes only pure variable costs? Vice...
-
What are some ways to forecast sales effectively for a retail business? For a manufacturer? For a service business?
-
Identify common factors that can lead to host compromise. Which factors are controllable by the host? Which factors are not controllable by the host?
-
Discuss the various systems for providing insurance to high-risk drivers
-
Solstice Company determines on October 1 that it cannot collect $ 50,000 of its accounts receivable from its customer P. Moore. Apply the direct write-off method to record this loss as of October 1.
-
Waterway Industries planned to use 1 yard of plastic per unit budgeted at $76 a yard. However, the plastic actually cost $75 per yard. The company actually made 4100 units, although it had planned to...
-
The muscle isozyme of lactate dehydrogenase is inhibited by lactate. Steady-state kinetic analysis yielded the following data, with lactate either absent or present at a fixed concentration. (a)...
-
(a) The plasmid pBR322 (4362 base pairs) was isolated and the circular DNA was found to be underwound, with a superhelix density () of -0.05. How many superhelical turns does this molecule have? (b)...
-
Suppose there is exactly one packet switch between a sending host and a receiving host. The transmission rates between the sending host and the switch and between the switch and the receiving host...
-
The present worth of an increasing geometric gradient is $23,632. The interest rate is 6% per year and the rate of change is 4% per year. The cash flow in year 1 is $3,000. a) List the parameters. (2...
-
The following information pertains to the City of Williamson for 2 0 2 4 , its first year of legal existence. For convenience, assume that all transactions are for the general fund, which has three...
-
Read the assigned readings from the textbooks: "The Development of the Canon"- Lutheran Bible Commentary (LBC) pgs 155-164"How the Bible came to us"- LBC pgs 827-830 Define the word "Canon" as it is...
-
A farmer is currently growing wheat and plans to sell it in September next year. He needs to plan his budget now, because of upcoming expenses. Suppose that the futures price of wheat for September...
-
Write a swing and event-driven program to display the following GUI and events. [40 Points] Rotate Clockwise Rotate Counter Clockwise Ac
-
Surrounding the condenser with which one of the following would be most effective for changing steam to water? A. High-pressure steam B. Low-pressure steam C. Stationary water D. Circulating water
-
Anna, a high school counselor, devised a program that integrates classroom learning with vocational training to help adolescents at risk for school dropouts stay in school and transition to work...
-
Name two control mechanisms that play a role in glycogen biosynthesis. Give an example of each.
-
Briefly outline the role of UDPG in glycogen biosynthesis.
-
Why is it advantageous that breakdown of glycogen gives rise to glucose-6-phosphate rather than to glucose?
-
Use the following data for problems #1 & #2: Belsky Corporation manufactures x-ray machines and has provided the following data from its activity-based costing system: Activity Cost Pool Total Cost...
-
The Sneed Corporation issues 12,000 shares of $46 par preferred stock for cash at $61 per share. The entry to record the transaction will consist of a debit to Cash for $732,000 and a credit or...
-
Suppose Seafood House restaurant is considering whether to (1) bake bread for its restaurant in-house or (2) buy the bread from a local bakery. The chef estimates that variable costs of making each...

Study smarter with the SolutionInn App


