Figure P 41.41 shows the first few energy levels of the lithium atom. Make a table showing
Question:
Figure P 41.41 shows the first few energy levels of the lithium atom. Make a table showing all the allowed transitions in the emission spectrum. For each transition, indicate
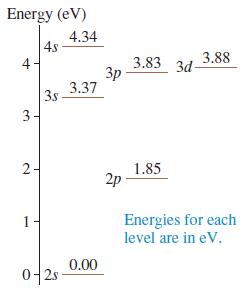
a. The wavelength, in nm.
b. Whether the transition is in the infrared, the visible, or the ultraviolet spectral region.
c. Whether or not the transition would be observed in the lithium absorption spectrum.
Transcribed Image Text:
Energy (eV) 4.34 4s 3.83 3d- 3p 3.37 3.88 4 3s 3- 2- 1.85 2p Energies for each level are in eV. 1 0.00 0-2s-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Visualize ab and c Solve We need to use the condition l 1 ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Physics For Scientists And Engineers A Strategic Approach With Modern Physics
ISBN: 9780321740908
3rd Edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Mathematics questions
-
Figure P 41.42 shows a few energy levels of the mercury atom. a. Make a table showing all the allowed transitions in the emission spectrum. For each transition, indicate the photon wavelength, in nm....
-
Make a table showing comparison b/w physical and chemical durability of concrete.
-
Make a table showing the sign conventions for mirrors and lenses. Include the sign convention for the mirrors and lenses themselves and for the image and object heights and distances for each.
-
A particular leadcadmium alloy is 8.0% cadmium by mass. What mass of this alloy, in grams, must you weigh out to obtain a sample containing 7.25 x 10 23 Cd atoms?
-
Repeat Prob. 1-112 for a pressure gage reading of 180 kPa.
-
To determine the Direct Labor Time Variance, the actual hours worked was 03 and the standard hours expected for the unit produced was 04, the standard rate per hour is $10.11, then the direct labor...
-
The sunspot cycle. Some plots against time show cycles of up-anddown movements. Figure 10.17 is a line graph of the average number of sunspots on the suns visible face for each month from 1900 to...
-
1. How would you characterize the main economic, legal-political, and sociocultural difference influencing the relationship between the partners in Shui Fabrics? What GLOBE Project dimensions would...
-
Joetz Corporation has gathered the following data on a proposed investment project (Ignore income taxes.): Investment required in equipment $ 30,000 Annual cash inflows $ 6,000 Salvage value of...
-
Write a program that calculates and displays values for y when y = xz / (x-z) your program should calculate y for values of x ranging between 1 and 5 and values of z ranging between 2 and 6. The...
-
Suppose you put five electrons into a 0.50-nm-wide one dimensional rigid box (i.e., an infinite potential well). a. Use an energy-level diagram to show the electron configuration of the ground state....
-
The ionization energy of an atom is known to be 5.5 eV. The emission spectrum of this atom contains only the four wavelengths 310.0 nm, 354.3 nm, 826.7 nm, and 1240.0 nm. Draw an energy-level diagram...
-
Repeat Prob. 341 for a location at an elevation of 1500 m where the atmospheric pressure is 84.5 kPa and thus the boiling temperature of water is 95C.
-
Management is what tradition used to call a liberal art: "liberal" because it deals with the fundamentals of knowledge, self-knowledge, wisdom, and leadership; "art" because it is a practice and...
-
Draft a five hundred and twenty five- to seven hundred-word internal communication planthat appropriately details your proposed solution to the internal team at CVS PHARMACY. In your communication...
-
Christopher Awnings was founded by Christopher Aminim in the early days of the retirement boom in the Okanagan to build and install custom retractable awnings for retirees to keep the sun out of the...
-
Leaders are responsible for making decisions that have long-term ramifications; thus, making the appropriate decisions can be stressful and leaders' decisions may vary. They often enhance employee...
-
Employee longevity A large insurance company has developed a model to identify the factors associated with employee turnover. The dependent variable is number of years an employee stays with the...
-
Use a web browser to go to the Gallup Organizations home page (http://www.gallup.com). The Gallup home page changes regularly. However, it should provide an opportunity to read the results of a...
-
A number of years ago the United Food and Commercial Workers Union organized 800 workers of the 1035 employees at one of the Wilson Brothers food operations in Toronto, Ontario. The employees include...
-
Two 500 g blocks of wood are 2.0 m apart on a frictionless table. A 10 g bullet is fired at 400 m/s toward the blocks. It passes all the way through the first block, then embeds itself in the second...
-
Two 500 g blocks of wood are 2.0 m apart on a frictionless table. A 10 g bullet is fired at 400 m/s toward the blocks. It passes all the way through the first block, then embeds itself in the second...
-
A 100 g granite cube slides down a 40 frictionless ramp. At the bottom, just as it exits onto a horizontal table, it collides with a 200 g steel cube at rest. How high above the table should the...
-
An underlying asset price is at 100, its annual volatility is 25% and the risk free interest rate is 5%. A European call option has a strike of 85 and a maturity of 40 days. Its BlackScholes price is...
-
Prescott Football Manufacturing had the following operating results for 2 0 1 9 : sales = $ 3 0 , 8 2 4 ; cost of goods sold = $ 2 1 , 9 7 4 ; depreciation expense = $ 3 , 6 0 3 ; interest expense =...
-
On January 1, 2018, Brooks Corporation exchanged $1,259,000 fair-value consideration for all of the outstanding voting stock of Chandler, Inc. At the acquisition date, Chandler had a book value equal...

Study smarter with the SolutionInn App


