Prove that the normalization constant of the 2p radial wave function of the hydrogen atom is (24a
Question:
Prove that the normalization constant of the 2p radial wave function of the hydrogen atom is (24πaB3)–1/2, as shown in Equations 41.7.
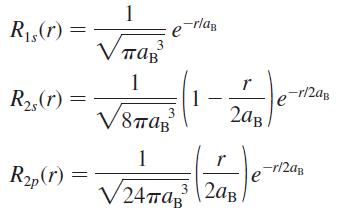
Transcribed Image Text:
1 R1,(r) = -rlag 1 ap -r/2ag R2,(r) 1 3 2ag 1 r R„(r) -r/2ag e V24ma, |2aB. 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
Solve From Equation 417 the 2p radia...View the full answer

Answered By

Khurram shahzad
I am an experienced tutor and have more than 7 years’ experience in the field of tutoring. My areas of expertise are Technology, statistics tasks I also tutor in Social Sciences, Humanities, Marketing, Project Management, Geology, Earth Sciences, Life Sciences, Computer Sciences, Physics, Psychology, Law Engineering, Media Studies, IR and many others.
I have been writing blogs, Tech news article, and listicles for American and UK based websites.
4.90+
5+ Reviews
17+ Question Solved
Related Book For 

Physics For Scientists And Engineers A Strategic Approach With Modern Physics
ISBN: 9780321740908
3rd Edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Mathematics questions
-
Prove that the normalization constant of the 1s radial wave function of the hydrogen atom is (a B 3 ) 1/2 , as given in Equations 41.7. A useful definite integral is
-
Prove that the normalization factor given by (12.2.12) ensures that (12.2.19) issatisfied. M-1 Ew?(n) (12.2.12) W(f)df = 1 J-1/2 (12.2.19)
-
The wave function for a hydrogen atom in the 2s state is(a) Verify that this function is normalized.(b) In the Bohr model, the distance between the electron and the nucleus in the n = 2 state is...
-
Contact local employers and ask for copies of their employee handbooks. If none are available, research parent companies of local employers online to see if their employee handbooks are available....
-
Determine the maximum amount of load, in kg, the balloon described in Prob. 1-102 can carry.
-
i solved most of the problem but i am still stuck 1 09 DO Rizio Co purchases a machine for $12.500, terms 2/10, 1/60. FOB shipping point. Rizio pold within the discount period and took the $250...
-
Define a marketing strategy.
-
a. Martha Painter, a PA, was appointed as the trustee of the So family trust. The So family trust owned the shares of the So Manufacturing Company, which is audited by another partner in Marthas...
-
5. Lon Corporation had beginning inventory of 42.000 units and expects sales of 96.000 units during the year. Desired ending Inventory is 31,000 units. How many units should Lon Corporation produce...
-
A company produces several products which pass through the two production departments in its factory. These two departments are concerned with filling and sealing operations. There are two service...
-
Prove that the radial probability density peaks at r = a B for the 1s state of hydrogen.
-
For an electron in the 1s state of hydrogen, what is the probability of being in a spherical shell of thickness 0.010a B at distance (a) 1/2 a B (b) a B (c) 2a B from the proton?
-
Preparing a Cash Flow Statement Manchester Corporation reported the following transactions and changes in account balances during the year ended December 31, 2000: Net income for 2000 was $160,000....
-
2 4. A rod of length 2 cm makes an angle rad with the principal axis of a thin convex lens. The lens has a focal 3 40 3 cm from the object as shown in the figure. The height of the length of 10 cm...
-
7. Consider an LC circuit, with inductance L = 0.1 H and capacitance C = 103 F, kept on a plane. The area of the circuit is 1 m. It is placed in a constant magnetic field of strength Bo which is...
-
Beach 10. The figure shows a circuit having eight resistances of 10 each, labelled R1 to R8, and two ideal batteries with voltages & = 12 V and 2 = 6 V. 1 R B1 Rs R R Which of the following...
-
12. Three plane mirrors form an equilateral triangle with each side of length L. There is a small hole at a distance /> O from one of the corners as shown in the figure. A ray of light is passed...
-
15. A small circular loop of area A and resistance R is fixed on a horizontal xy-plane with the center of the loop always on the axis of a long solenoid. The solenoid has m turns per unit length and...
-
On March 1, 20Y1, Sadie May established Pampered Pet, which completed the following transactions during the month: a. Sadie May transferred cash from a personal bank account to an account to be used...
-
What is the difference between direct materials and indirect materials?
-
The nucleus of the polonium isotope 214 Po (mass 214 u) is radioactive and decays by emitting an alpha particle (a helium nucleus with mass 4 u). Laboratory experiments measure the speed of the alpha...
-
A neutron is an electrically neutral subatomic particle with a mass just slightly greater than that of a proton. A free neutron is radioactive and decays after a few minutes into other subatomic...
-
A neutron is an electrically neutral subatomic particle with a mass just slightly greater than that of a proton. A free neutron is radioactive and decays after a few minutes into other subatomic...
-
Domino is 4 0 years old and is married out of community of property with the exclusion of the accrual system to Dolly ( 3 5 ) . They have one child, Domonique, who is 1 1 years old. Domino resigned...
-
YOU ARE CREATING AN INVESTMENT POLICY STATEMENT FOR JANE DOE General: 60 years old, 3 grown children that are living on their own and supporting themselves. She is in a very low tax rate so we don't...
-
firm purchased a new piece of equipment with an estimated useful life of eight years. The cost of the equipment was $65,000. The salvage value was estimated to be $10,000 at the end of year 8. Using...

Study smarter with the SolutionInn App


