Question: Use the data in problem 8.29 to compute the partial molar enthalpies of pyridine and acetic acid in their mixtures at 25C over the whole
Use the data in problem 8.29 to compute the partial molar enthalpies of pyridine and acetic acid in their mixtures at 25°C over the whole composition range.
Problem 8.29
Two streams containing pyridine and acetic acid at 25°C are mixed and fed into a heat exchanger. Due to the heat-of-mixing effect, it is desired to reduce the temperature after mixing to 25°C using a stream of chilled ethylene glycol as indicated in the diagram. Calculate the mass flow rate of ethylene glycol needed. The heat capacity of ethylene glycol at these conditions is approximately 2.8 kJ/(kg K), and the enthalpy change of mixing (ΔmixH) is given below.
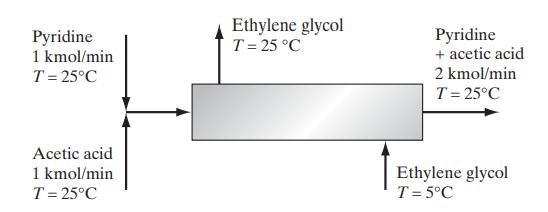
Data: Heat of mixing for pyridine (C5H5N) and acetic acid at 25°C [H. Kehlen, F. Herold and H.- J. Rademacher, Z. Phys. Chem. (Leipzig), 261, 809 (1980)].
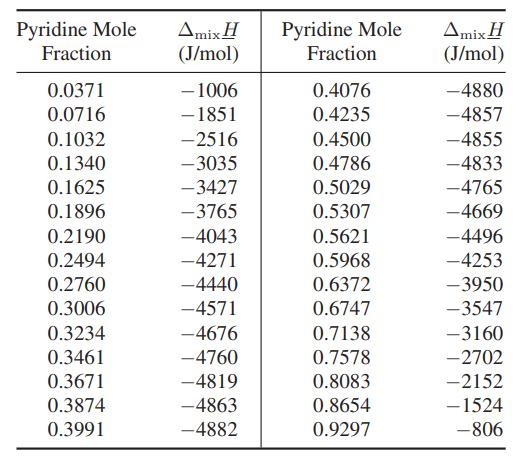
Pyridine 1 kmol/min T = 25C Acetic acid 1 kmol/min T = 25C Ethylene glycol T = 25 C Pyridine + acetic acid 2 kmol/min T = 25C Ethylene glycol T = 5C
Step by Step Solution
3.45 Rating (161 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


