Identify the reactions with K > 1 among the following reactions and, for each such reaction, write
Question:
Identify the reactions with K > 1 among the following reactions and, for each such reaction, write balanced reduction and oxidation half-reactions. For those reactions, show that K > 1 by calculating the standard Gibbs free energy of the reaction. Use the smallest whole-number coefficients to balance the equations.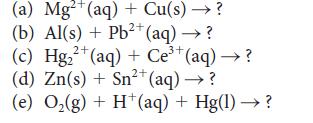
Transcribed Image Text:
(a) Mg²+ (aq) + Cu(s) → ? (b) Al(s) + Pb²+ (aq) → ? 2+ 2+ (c) Hg₂+ (aq) + Ce³+ (aq) → ? (d) Zn(s) + Sn²+ (aq) → ? (e) O₂(g) + H+ (aq) + Hg(1)→→?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To identify the reactions with K 1we need to calculate the standard Gibbs free energy G of each reac...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The oxidation of SO 2 to SO 3 is one of the reactions involved in the formation of acid rain. If you want to predict the spontaneous direction of the reaction for a specific mixture of the gases, you...
-
1. Given Pascal's Triangle 4 1 3 1 2 6 1 3 1 5 10 10 4 1 5 1 1 a. Explain how the n and r values for C, relate to the number 6 shown above in Pascal's triangle. Justify your explanation by showing...
-
Many important biological reactions involve electron transfer. Because the pH of bodily fluids is close to 7, the biological standard potential of an electrode, E*, is measured at pH = 7....
-
What is a safe edge on a file?
-
Balanced scorecard Following is a random-order listing of perspectives, strategic objectives, and performance measures for the balanced scorecard. For each perspective, select those strategic...
-
The premise of the Planet of the Apes movies and book is that hibernating astronauts travel far into Earth's future, to a time when human civilization has been replaced by an ape civilization....
-
What are the benefits and drawbacks of starting most new hires at the help-desk function? The IT Department at Hamelin Hospital Hamelin Hospital is a large (700-bed) regional hospital in the...
-
Olathe Company exchanges old delivery equipment for new delivery equipment. The book value of the old delivery equipment is $31,000 (cost $61,000 less accumulated depreciation $30,000). Its fair...
-
A firm has 50,000 shares of stock outstanding, net income of $50,000, and a PE ratio of 10. What will the firms PE ratio be if the firm repurchases 25,000 shares? Assume all else remains constant....
-
Moravanti Italian Imports has four employees and pays biweekly. Assume that box 2 is not checked for L. Torabi and R. Beninati and is checked for G. Fisher and J. Tillman. Required: Complete the W-4...
-
Suppose that 30.0 mL of 0.12 m C 6 H 5 COOH(aq) is titrated with 0.20 m KOH(aq). (a)What is the initial pH of the 0.20 m C 6 H 5 COOH(aq)? (b)What is the pH after the addition of 5.00 mL of 0.20 m...
-
Draw the Lewis structure or symbol for each of the following species and identify each one as a Lewis acid or Lewis base: (a) SO; (b) I; (c) CH3S (the C atom is the central atom); (d) NH; (e) NO.
-
The case in Exercise 2. For the following joint distributions, find the covariance of X and Y using the computational method, Cov(X, Y) = E(XY) - . 1 0.45 0.25 Y=3 0.05 0.25
-
Directions: Put your feet in the shoes of the business owner and suggest specific ways on how a business can gain profit and how it can be avoid loss Ways to Gain Profit 1. 2. 3. 4. 5. 1. 2. 3. 4. 5....
-
Define business intelligence Briefly discuss how your organisation can use business intelligence to improve decision-making Please see below rubric as guidance. Kindly list references in APA 7th...
-
Mr Santos apply for college educational plan for his 3 children .The 3 children ages are 6 yrs old , 3 yrs old and 1 yr old. The fund will be set-up the deposit of a fixed sum on the child's current...
-
Recovery Centers of America needs to acquire new vehicles that will cost $2.5 million across its six state service area. It plans to use the vehicles for three years, at which time new vehicles will...
-
The following program is supposed to allow the user to enter a list of numbers, then print them out formatted with 2 decimal positions. However, there are 3 errors. Indicate the line number of each...
-
The following normal quantile plot shows the distribution of the diameters, in cm, of each of nine American Sycamore trees. 34 The normal quantile plot is not linear, which suggests that a...
-
Use nodal analysis to determine voltages v1, v2, and v3 in the circuit Fig. 3.76. Figure 3.76 4 S 3i, 2 A 4A
-
Benzyl acetate is a pleasant-smelling ester found in the essential oil of jasmine flowers and is used in many perfume formulations. Starting with benzene and using any other reagents of your choice,...
-
Aspartame (below) is an artificial sweetener used in diet soft drinks and is marketed under many trade names, including Equal TM and Nutrasweet TM . In the body, aspartame is hydrolyzed to produce...
-
Draw a plausible mechanism for each of the following transformations: a. b. c. d. e. Pyridine CI
-
Please help me answer all these question one by one. THE COMPANY IS GOING TO BE WALMART (PLEASE DO NOT USE AND PREVIOUS STUDENT ANSWERS) Remember, one-page memo first. search the web for these...
-
keAssignmentMain.do?invoker=&takeAssignmentSessionLocator=&inprogress=false Print Item Using the data below for the Ace Guitar Company: 6 A Region Sales B Region $500,000 Cost of goods sold $900,000...
-
please answer both parts Winston Company estimates that total factory overhead for the following year will be $979,200. The company has decided that the basis for applying factory overhead should be...

Study smarter with the SolutionInn App


