Consider the reaction: Calculate G rxn for the reaction at 25 C under each of the following
Question:
Consider the reaction:
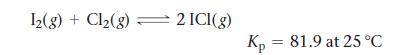 Calculate ΔGrxn for the reaction at 25 °C under each of the following conditions:
Calculate ΔGrxn for the reaction at 25 °C under each of the following conditions:
a. Standard conditions
b. At equilibrium
c. PICl = 2.55 atm; PI2 = 0.325 atm; PCl2 = 0.221 atm
Transcribed Image Text:
12(g) + Cl₂(g) = 2 ICI(g) Kp = 81.9 at 25°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The standard Gibbs free energy change AG ...View the full answer

Answered By

John Kago
Am a processional practicing accountant with 5 years experience in practice, I also happens to have hands on experience in economic analysis and statistical research for 3 years. am well conversant with Accounting packages, sage, pastel, quick books, hansa world, etc, I have real work experience with Strata, and SPSS
4.70+
31+ Reviews
77+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the reaction: Calculate G rxn for the reaction at 25 C under each of the following conditions: a. Standard conditions b. At equilibrium c. P CH3OH = 1.0 atm; P CO = P H2 = 0.010 atm CO(g) +...
-
Ray Holt Corporation has retained you as a consultant on accounting policies and procedures. During 2019, the company engaged in a number of treasury stock transactions, having foreseen an...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
What problems may be encountered in making a comparative study of remuneration reports?
-
What is the difference between an activity flexible budget and a functional-based (traditional) flexible budget?
-
The accompanying table shows the U.S. domestic demand schedule and domestic supply schedule for oranges. Suppose that the world price of oranges is $0.30 per orange. a. Draw the U.S. domestic supply...
-
C2.8. Price-to-book ratiosare determined byhowaccountants measure bookvalues. Can you think of accounting reasons for why price-to-book ratios were high in the 1990s? Whatotherfactors mightexplain...
-
Three point charges are arranged on a line. Charge q3 = + 5.00 nC and is at the origin. Charge q2 = - 3.00 nC and is at x = +4.00cm. Charge q1 is at x = +2.00cm. What is q1 (magnitude and sign) if...
-
8. On May 1, Charlie Co. sold 12,000 yearly subscriptions of its monthly magazine to customers for $40 each. On June 1, customers received their first subscription of the magazine. Prepare the...
-
Explain the difference between macrostates (external arrangements of particles) and microstates (internal arrangements of particles).
-
Estimate the value of the equilibrium constant at 525 K for each reaction in Problem 73. Problem 73 Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction....
-
What are reversing entries? When are reversing entries useful?
-
Based on a survey, assume that 42% of consumers are comfortable having drones deliver their purchases. Suppose that we want to find the probability that when six consumers are randomly selected,...
-
What is the social location that determines this speech community? Is it determined by race, class, gender, sexuality, or some other social location? What makes this speech community unique? What are...
-
Write a program named SumOfNumberOfSquares.java that prompts user to enter a number of integers and calculates the sum of their squares. The following is a sample run. The green fonts represent user...
-
6.4 Charles Augustin de Coulomb was a French physicist who is best known for formulating the law that calculates the force between two electric charges. To honor Coulomb, the unit of electric charge...
-
What amount of cash payments to suppliers will be reported by Indigo Company for the year ended December 31, 2024?
-
Advances in research and development in the pharmaceutical industry have enabled manufacturers to identify potential cures more quickly and therefore at lower cost. At the same time, the aging of our...
-
Repeat Exercise 16.6 using the t-test of the coefficient of correlation. Is this result identical to the one you produced in Exercise 16.6?
-
Name a type of compressor often used for pneumatic fluid power systems.
-
The intake duct to a fan consists of intake louvers, 5.8 m of square duct (800 800 mm), a sudden contraction to a 400-mm-diameter round duct, and 9.25 m of the round duct. Estimate the pressure at...
-
The electric field of an electromagnetic plane wave is given in SI units by (a) What is the wave?s angular frequency? (b) Write an expression for vector k. (c) What is the value of k? (d) Determine...
-
Which of the following programs covers custodial care? A HMOs B Medicare Part B C PPOs D Medicare Part A E Medicaid
-
uppose a taxpayer has exhausted his lifetime exclusion amount and has $14 million. a. Assuming a flat 40% gift tax rate, what is the maximum amount a taxpayer can transfer to her daughter (and still...
-
Physical Units Method, Relative Sales Value Method Farleigh Petroleum, Inc., is a small company that acquires high - grade crude oil from low - volume production wells owned by individuals and small...

Study smarter with the SolutionInn App


