Consider the structure of the amino acid aspartic acid. Indicate the hybridization about each interior atom. H
Question:
Consider the structure of the amino acid aspartic acid. Indicate the hybridization about each interior atom.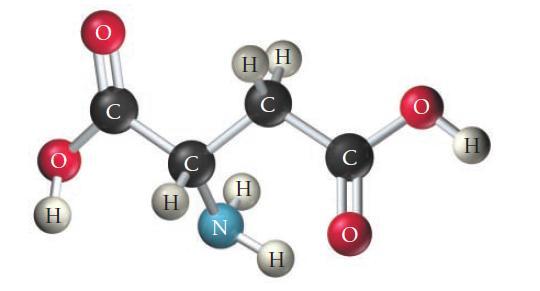
Transcribed Image Text:
H 0 0 Z H H H H О
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
OHCOCH2CNH2COOH this is the structure of aspartic acid The hybridization of c...View the full answer

Answered By

Jeetu sahu
I completed my B.Tech in Textile Engineering from NIT JALANDHAR,INDIA. I can help students to solve assignments and exams of physics ,mathematics, statistics, science and engineering. I am doing it since three years. I can provide tuition for Physics and Mathematics .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suppose you are the head of the marketing department for an on-demand video streaming service. A registered user pays a monthly subscription fee to access the platforms entire library. Your research...
-
Consider the structure of the amino acid alanine. Indicate the hybridization about each interior atom. H H H H
-
Write a hybridization and bonding scheme for each molecule that contains more than one interior atom. Indicate the hybridization about each interior atom. Sketch the structure, including overlapping...
-
Hi,Please answer no.5 and 6 only. This is my 3rd time to uploadthis question. Thank you so much! Bay Lake Mining Ltd. purchases earth-moving equipment on 1 August \( 20 \times 6 \) and signs a...
-
Information for Dilts Corporation is given in BE19-7. If the company has fixed costs of $199,500, how many units of each model must the company sell in order to break even?
-
Pam Huber worked at Wal-Mart as a grocery order filler, earning $13 an hour. While on the job, she suffered a permanent injury to her right arm and hand. Both she and Wal-Mart agreed that she was...
-
A machine shop has 8 identical drilling machines manned by 6 operators. The machines cannot be worked without an operator wholly engaged on it. The original cost of all these 8 machines works out to...
-
How important is it that the warehouse bins and invoices are coded inconsistently? Respond in a paragraph. What are some of the problems created when a code appears to be mnemonic but employees are...
-
A company's $100 par value preferred stock with a dividend rate of 15% per year is currently priced at $105.85 per share. The company's earnings are expected to grow in an annual rate of 3% for the...
-
Sketch the bonding molecular orbital that results from the linear combination of two 1s orbitals. Indicate the region where interference occurs and state the kind of interference (constructive or...
-
Write a hybridization and bonding scheme for each molecule or ion. Sketch the structure, including overlapping orbitals, and label all bonds using the notation shown in Examples 11.6 and 11.7. a. SO...
-
Why might a low-income country put up barriers to trade, such as tariffs on imports?
-
As a new principal, I assigned a teacher to a different grade for the coming year. I did not expect to cause the anxiety it did. The teacher first came to me in tears and begged for her assignment to...
-
Peruse the following websites to learn about the different ways of categorizing leadership. 1. https://www.businessnewsdaily.com/9789-leadership-types.html 2....
-
Making Consumer Choices The Espresso Machine (25 points) In real life, you must often make choices about whether to buy something pre-made or make it yourself. There are many things to consider:...
-
1) Read over the article/case and summarize what it is referring to in your own words. 2) What type of leadership traits can you describe in the case study? Use materials both from the handout and...
-
After reading or watching, https://smallbusiness.chron.com/internal-analysis-important-80513.html https://www.indeed.com/career-advice/career-development/internal-analysis...
-
You are considering making a loan to Kellogg's. The following information is from the statements of cash flows and the notes to the consolidated financial statements included in Form 10-K for fiscal...
-
The population of Detroit, Michigan, decreased from 1,027,974 in 1990 to 688,701 in 2013 (Source: U.S. Census Bureau). Find the average rate of change in the population of Detroit, Michigan, over the...
-
What does the term ISA mean? Do the Pentium 4 -> IBM POWER7 and Pentium 3 -> IBM POWER8 have the same ISA?
-
The diameter of an atom is roughly 104 times the diameter of its nucleus. If the nucleus of an atom were 1 mm across, how many feet across would the atom be?
-
In Section 10.1.3 we talked about introducing an intrinsic phase shift ε between oscillators in a linear array. With this in mind, show that Eq. (10.18) becomes when the incident plane...
-
ABC Corporation has an activity - based costing system with three activity cost pools - Machining, Setting Up , and Other. The company's overhead costs, which consist of equipment depreciation and...
-
Consolidated Balance Sheets - USD ( $ ) $ in Thousands Dec. 3 1 , 2 0 2 3 Dec. 3 1 , 2 0 2 2 Current assets: Cash and cash equivalents $ 9 8 , 5 0 0 $ 6 3 , 7 6 9 Restricted cash 2 , 5 3 2 Short -...
-
How does corporate governance contribute to investor confidence and stakeholder trust? Accounting

Study smarter with the SolutionInn App


