Determine the product of the reaction: CH3-CH-CH-OH CH3 a) CH3-CH - CH3 Cl b) CH3-CH - CH3
Question:
Determine the product of the reaction: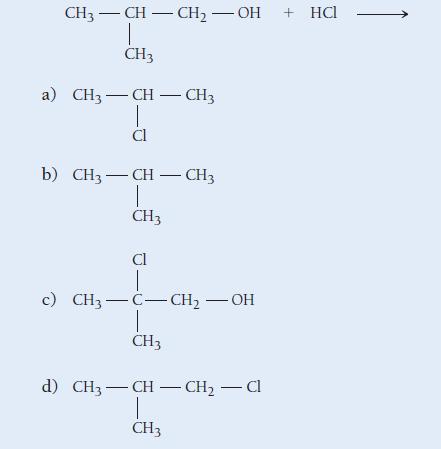
Transcribed Image Text:
CH3-CH-CH₂-OH CH3 a) CH3-CH - CH3 Cl b) CH3-CH - CH3 I CH3 CI c) CH3–C–CH, - OH CH3 d) CH3-CH-CH₂ - Cl | CH3 + HCl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
d C...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Demonstrate your knowledge of palladium cross-coupling reactions by suggesting structures for compounds (14) and (15). Suggest reagent(s) that would facilitate the cross-coupling reaction between...
-
When (S)-1-bromo-1-fluoroethane reacts with sodium methoxide, an S N 2 reaction takes place in which the bromine atom is replaced by a methoxy group (OMe). The product of this reaction is...
-
Sodium-23 (in a sample of NaCl) is subjected to neutron bombardment in a nuclear reactor to produce 24 Na. When removed from the reactor, the sample is radioactive, with activity of 2.54 10 4 dpm....
-
Do you think firms rely too much on internal funds? Would it be better if they had to go back to shareholders when they needed cash?
-
Indicate whether each of the following actions is related to (a) Managing cash needs, (b) Setting credit policies, (c) Financing receivables, or (d) Ethically reporting receivables: 1. Selling...
-
a. What is the linear model? b. Which two parameters define the population straight-line relationship? c. What sample statistics are used to estimate the three population parameters , , and ? d. Is...
-
Is leadership necessarily moral? Was Hitler a leader even though he was evil? Why or why not?
-
1. Develop a multiple regression equation using unemployment rate and average monthly temperature to predict emergency road service calls. 2. Create a new temperature variable and relate it to...
-
Solve the equation. x+4-1=0 Enter the solution in the box. Show Hints
-
Which compound is an ester? a) CH3 - CH -0 - CH3 O b) CH3-CH-C-OH c) CH3-C- CH3 O= d) CH3-C-0-CHCH3
-
Determine the product of the reaction: CH3 I CH3 -CH=C-CH3 + HBr CH3 T a) CHBr -CH=C-CH3 CH3 b) CH3 - CH -CH-CH3 CH3 c) CH3 -CH=CH-CH3 Br CH3 d) CH3 - CH -C-CH3 Br
-
On July 1, 2012, a Japanese company enters into a forward contract to buy \(\$ 1\) million with yen on January 1, 2013. On September 1, 2012, it enters into a forward contract to sell \$1 million on...
-
Prepare a statement of financial position, statement of comprehensive income and statement of changes in equity in accordance with International Financial Reporting Standards.
-
When one thinks about the spirit of Britain, especially London, Links of London is hoping that their charm, elegance, and world-renowned reputation make them the firm that immediately comes to the...
-
Explain, using an appropriate diagram, how a deflationary gap can occur and how this gap can be eliminated.
-
1. E-Z Open Manufacturing Company is a leading maker of manual can openers. In the year just closed, E-Z Open controlled 17.2% of the manual can opener market in North America. That placed the...
-
The manager of Swift Car Inspection reviewed his monthly operating costs for the past year. His costs ranged from \($4,400\) for 1,400 inspections to \($4,000\) for 900 inspections. Requirements 1....
-
Suppose a manager wants to borrow $50 million of a Treasury security that it plans to purchase and hold for 20 days. The manager can enter into a reverse repo agreement with a dealer firm that would...
-
The swap spread is the difference between the swap rate and the equivalent-maturity Treasury bond yield. Explain why a widening swap spread may be a signal of deteriorating economic conditions. Plot...
-
A rod 50 cm long and 1.0 cm in radius carries a 2.0C charge distributed uniformly over its length. Find the approximate magnitude of the electric field (a) 4.0 mm from the rod surface, not near...
-
Whats the approximate field strength 1 cm above a sheet of paper carrying uniform surface charge density s = 45 nC/m 2 ?
-
What is the electric field strength just outside the surface of a conducting sphere carrying surface charge density 1.4 C/m 2 ?
-
In evaluating the financial performance of a business that is for sale, the potential new owner should compare financial ratios to: Multiple Choice their business plan. their largest competitor....
-
The following day you receive a $600 cash payment for a two week (14 days) rental period, starting the 8th of February 2022. This payment is received from Paul Stone, 2 Watchtower Way, Torquay. There...
-
Gastow Pumps is a manufacturer of commercial and heavy industrial Pumps. The firm's two product lines are called Directlift and Gravity. The primary raw materials are flexible steel sheets, and 23cm...

Study smarter with the SolutionInn App


