Find G rxn for the reaction: Use the following reactions with known G rxn values: 3 C(s)
Question:
Find ΔG°rxn for the reaction:![]()
Use the following reactions with known ΔG°rxn values: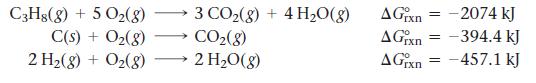
Transcribed Image Text:
3 C(s) + 4H₂(g) → C3H8(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To work this problem you need to manipulate the given reactions with known values of AGxn in such a ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following reaction taking place at 300 K: 2 NH 3 ( g ) + 3 Br 2 ( g ) N 2 ( g ) + 6 HBr ( g ) K c = ?? a) Use the following reactions with known values of K c to determine the value...
-
Proper management and leadership are key when dealing with finances in sports. Explain the difference between the two and how they will apply to the financial structure of your sports organization?
-
Calculation of H rxn for Part I 0 1. Assume the heat capacity of the final solution is 4.184 J K-1 g-1. Using the final mass of the solution in the calorimeter, calculate q contents from equation...
-
In the figure below, a square of edge lengths is formed by four spheres of masses, m, M, m3, and m4. What is the x component and the y component of the net gravitational force from them on a central...
-
Tilly Issac is the assistant controller for Tagwell Corporation, a leading producer of home appliances. Her friend Zack Marsh is the supervisor of the firms Cookware Department. Marsh has the...
-
A corporate customer borrows $ 150,000 against the firms credit line at a local bank. Indicate with a T- account how the transaction will affect the banks deposit balances held at the Federal Reserve...
-
When oil and gas are produced jointly, what basis or units of measure are allowed for full cost amortization? When oil and gas are produced jointly, what basis or units of measure are allowed for...
-
The adjusted trial balance for Anara Co. as of December 31, 2013, follows. P. Anara invested $40,000 cash in the business during year 2013 (the December 31, 2012, credit balance of the P. Anara,...
-
Question 23 Points 1 Find ZQRS if the lines containing the line segments PQ and RS are parallel. 60 90 120 180 120 P 95 S R
-
A reaction is spontaneous under a certain set of conditions. What can you conclude about G rxn and Q? (a) AGxn < 0, Q > K (c) AGrxn > 0, Q < K (b) AGxn <0, Q < K (d) AGxn> 0, Q > K
-
Why does the entropy of a gas increase when it expands into a vacuum?
-
In Exercises 49 through 52, the graph of a derivative function y = f'(x) is given. Describe the function f (x), and sketch a possible graph of y = f (x). -3 + 1 y = f'(x) 5 -X
-
On January 6, 1992, the growing controversy over the safety factor led the U.S. Food and Drug Administration to call for a moratorium on breast implants.1 As January wore on, the crisis deepened...
-
It was early on a Friday morning in London7:15 a.m. on February 24, 1995, to be exactthat the phone call came for Peter Baring from Peter Norris. Barings family had been in banking since 1763. They...
-
Parental consent for a medical or surgical procedure on a minor is generally: Required prior to the procedure. Not required if the minor is married or otherwise emancipated.
-
Regardless of religious beliefs, patients have the right to refuse medical treatment. If a patient refuses treatment: The refusal should be noted in the patients medical record. A release form...
-
On September 30, 2004, Merck voluntarily withdrew its rheumatoid arthritis drug (Vioxx) from the market due to severe adverse effects observed in many of its users (Exhibit 1). As a result, Mercks...
-
Gildan Activewear Inc. is a Canadian company that manufactures and sells activewear, socks, andunderwear. Manufacturing is primarily done in Honduras and Dominican Republic and sales are made...
-
Reconsider Prob. 1474. In order to drain the tank faster, a pump is installed near the tank exit as in Fig. P1475. Determine how much pump power input is necessary to establish an average water...
-
Sound waves in water travel at a speed of approximately 1500 m/s. What is the wavelength of a sound wave that has a frequency of 440 Hz?
-
The neck of a guitar is designed with frets as shown in Figure Q12.8. A player can hold a string against one of the frets and thus shorten the vibrating length L of the string. In this way, a...
-
A wave on a Slinky has a frequency of 2.3 Hz. It takes 3.5 s to travel the length of the Slinky (0.30 m). What is the wavelength?
-
Problem 9-12 Direct Materials and Direct Labor Budgets [LO4, LO5] The production department of Prison Company has submitted the following forecast of units to be produced by quarter for the upcoming...
-
Exercise 10-6 (Algo) Contrasting Return on Investment (ROI) and Residual Income [LO10-1, LO10-2] Meiji Isetan Corporation of Japan has two regional divisions with headquarters in Osaka and Yokohama....
-
The current sections of Bramble Inc's balance sheets at December 31, 2019 and 2020, are presented here. Bramble's net income for 2020 was $207,500. Depreciation expense was $34,000. 2020 2019 Current...

Study smarter with the SolutionInn App


