The first six elements of the first transition series have the following number of stable isotopes: Explain
Question:
The first six elements of the first transition series have the following number of stable isotopes: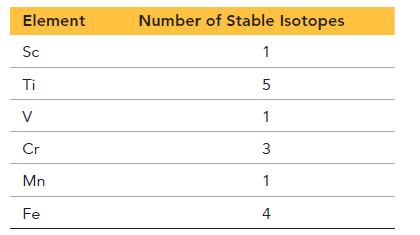
Explain why Sc, V, and Mn each have only one stable isotope while the other elements have several.
Transcribed Image Text:
Element Sc Ti V Cr Mn Fe Number of Stable Isotopes 1 5 1 3 1 4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Sc V and Mn each have odd numbe...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Which of the following questions involve microeconomics, and which involve macroeconomics? In each case, explain your answer. a. Why did consumers switch to smaller cars in 2008? b. Why did overall...
-
Explain how costs are assigned to activities.
-
Consider Figure 1-14. Explain the meaning of the line that connects CUSTOMER to ORDER and the line that connects ORDER to INVOICE. What does this say about how Pine Valley Furniture Company does...
-
E2.8. Finding Financial Statement Information on the Internet (Easy) The Securities and Exchange Commission (SEC) maintains the EDGAR database of com- pany filings with the commission. Explore the...
-
The standard cost of Product B manufactured by TLC Company includes three units of direct materials at $5.00 per unit. During June, 28,000 units of direct materials are purchased at a cost of $4.70...
-
Burnese Mountain Inc. manufactures two products, Product C and Product D. The company estimated it would incur $130,890 in manufacturing overhead costs during the current period. Overhead currently...
-
Suppose that an 85.0-gram laboratory animal ingests 10.0 mg of a substance that contained 2.55% by mass Pu-239, an alpha emitter with a half-life of 24,110 years. a. What is the animals initial...
-
Calculate the quantity of energy produced per mole of U-235 (atomic mass = 235.043922 amu) for the neutron-induced fission of U-235 to produce Te-137 (atomic mass = 136.9253 amu) and Zr-97 (atomic...
-
Mohit Corp. is a manufacturer of specialized diving gear. What internal control could be tested by its auditor to support the objective of proper period cutoff for their Cost of Goods Sold accounts?...
-
Claxton, Inc. paid a dividend of $ 0 . 9 5 per common share every December from 2 0 0 9 through 2 0 2 3 . The dividend is expected to continue at that level in 2 0 2 4 and 2 0 2 5 . In 2 0 2 6 and...
-
1. Who are two (2) specific examples of effective leaders (who you know personally) who have impacted your life? 2. What made them "effective" leaders? What many specific traits did these leaders...
-
pr Hwk12 Consider the differebtiable function F(x) whose instantaneous rates are given by the table of values below: I 3.00 3.50 4.00 4.50 5.00 5.50 6.00 6.50 7.00 F'(x) 21.00 27.50 35.00 43.50 53.00...
-
A stone was dropped off a cliff and hit the ground with a speed of 152 ft/s. What is the height of the cliff? (Use 32 ft/s for the acceleration due to gravity.) Step 1 We know that s(t) = 1 at + vot...
-
The 150 m long beam is submitted to a distributed load w(x) = (0.05 x 2) + 10 N/m. 50 50 w(x) 100 150 What is the moment about the point O in kN.m created by the distributed load? O-25.7 kN.m O-249...
-
If the price of processor chips used in manufacturing personal computers decreases, what will happen in the market for personal computers? How will the equilibrium price and equilibrium quantity of...
-
1. What are some current issues facing Saudi Arabia? What is the climate for doing business in Saudi Arabia today? 2. Is it legal for Auger's firm to make a payment of $100,000 to help ensure this...
-
Why is gravity considered a fundamental force whereas the force a bat exerts on a ball is not?
-
Of the following, the shortest is a. 1 mm b. 0.01 in. c. 0.001 m d. 0.001 ft
-
What is the difference between spring and neap tides? Under what circumstances does each occur?
-
Assignment Title: The Role of Bookkeeping in Business Management and Financial Reporting Objective: Understand the importance of proper bookkeeping procedures in the management of...
-
17) The adjustment that is made to allocate the cost of a building over its expected life is called:A) depreciation expense.B) residual value.C) accumulated depreciation.D) None of the above answers...
-
9) Prepaid Rent is considered to be a(n):A) liability.B) asset.C) contra-asset.D) expense.10) As Prepaid Rent is used, it becomes a(n):A) liability.B) expense. C) contra-asset.D) contra-revenue.11)...

Study smarter with the SolutionInn App


