The mass of fuel in a jet must be calculated before each flight to ensure that the
Question:
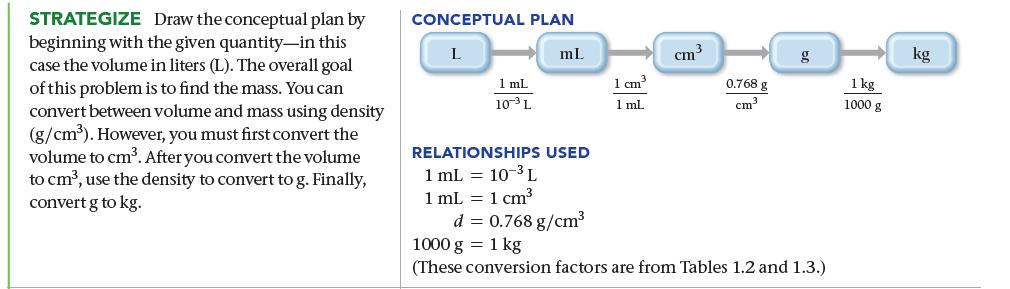
The mass of fuel in a jet must be calculated before each flight to ensure that the jet is not too heavy to fly. A 747 is fueled with 173,231 L of jet fuel. If the density of the fuel is 0.768 g/cm3, what is the mass of the fuel in kilograms?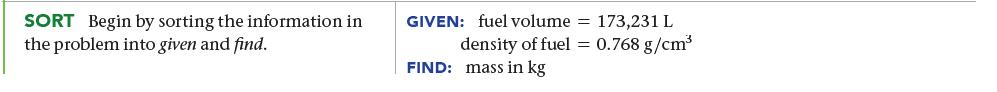
Transcribed Image Text:
SORT Begin by sorting the information in the problem into given and find. GIVEN: fuel volume = 173,231 L density of fuel = 0.768 g/cm³ FIND: mass in kg
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
173231 L X 1 ...View the full answer

Answered By

Benish Ahmad
I'm a professional software engineer. I'm lectutrer at GCUF and I have 3 years of teaching experience. I'm looking forward to getting mostly computer science work including:
Programming fundamentals
Object oriented programming
Data structures
object oriented design and analysis
Database system
Computer networks
Discrete mathematics
Web application
I am expert in different computer languages such as C++, java, JavaScript, Sql, CSS, Python and C#. I'm also have excellent knowledge of essay writing and research. I have worked in other Freelancing website such as Fiverr and Upwork. Now I have finally decided to join the SolutionInn platform to continue with my explicit work of helping dear clients and students to achieve their academic dreams. I deliver plagiarism free work and exceptional projects on time. I am capable of working under high pressure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Lakefield Corporations oil trading desk buys and sells oil products (crude oil and refined fuels), options, and futures in international markets. The trading desk is responsible for buying raw...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
A stationary jet engine is shown. Air with a density of 0.0805 lb/ft 3 enters as shown. The inlet and outlet cross sectional areas are both 10.8 ft 2 . The mass of fuel consumed is 2% of the mass of...
-
Graph the sets of points whose polar coordinates satisfy the equations and inequalitie. 0 , r = 1
-
Go to Case 21.3,Spray-Tek, Inc. v. Robbins Motor Transportation, Inc., 426 F.Supp.2d 875 (W.D. Wis. 2006), on pages 428430. Read the excerpt and answer the following questions. (a) Issue: What...
-
The city of Lydell expended federal awards from the following programs during 2019. Assume the auditor has given an unqualified opinion on the financial statements and reports no material weaknesses...
-
The Bell and Howell partnership is considering three long-term capital investment proposals. Each investment has a useful life of 5 years. Relevant data on each project are as follows. Depreciation...
-
The following graph shows the labor market for research assistants in the fictional country of Universalia. The equilibrium wage is $10 per hour, and the equilibrium number of research assistants is...
-
Ginos Restaurant is a popular restaurant in Boston, Massachusetts. The owner of the restaurant has been trying to better understand costs at the restaurant and has hired a student intern to conduct...
-
One criticism of Hofstedes work is that the scores on each dimension reflect only an average tendency of a particular country and, therefore, inadequately reflect the wide range of responses given...
-
What is the difference between a crystalline solid and an amorphous solid?
-
Explain the difference between a pure substance and a mixture
-
(a) If g(x) = (sin2x)/x2, use your calculator or computer to make a table of approximate values of for t = 2, 5, 10, 100, 1000, and 10,000. Does it appear that is convergent? (b) Use the Comparison...
-
Cecilia Company reported net income of $1,400,000. The average total liabilities were $4,310,000 and average total stockholders' equity was $5,220,000. Interest expense was $102,000 and the tax rate...
-
Assume that S(ILS/INR) = 0.0449 and that F(ILS/INR) = 0.0415 (ILS = Israeli New Shekel; INR = Indian Rupee). The interest rate in India is 4.71%, while the interest rate in Israel is 0.10%. Also...
-
A university spent $2 million to install solar panels atop a parking garage. These panels will have a capacity of 300 kilowatts (kW) and have a life expectancy of 20 years. Suppose that the discount...
-
Cool Fizzy Ltd's production manager Phil Palm has been under pressure from senior management to improve divisional profitability (which is also incidentally linked to his management performance...
-
What is the difference between a left outer join and a right outer join? Give an example.
-
The juror pool for the upcoming murder trial of a celebrity actor contains the names of 100,000 individuals in the population who may be called for jury duty. The proportion of the available jurors...
-
On 1 July 2021, Croydon Ltd leased ten excavators for five years from Machines4U Ltd. The excavators are expected to have an economic life of 6 years, after which time they will have an expected...
-
Calculate the energy in kJ necessary to vaporize 100 g of water at its boiling point.
-
Octane has a normal boiling point of 125.5 o C and a heat of vaporization (H vap ) of 38.8 kJ / mol. What is the vapor pressure of octane at 20 o C?
-
A form of iron crystallizes with a body-centered unit cell. The radius of iron is 124 pm. Calculate the density of this form of iron in g/cm 3 .
-
Jane provides a money purchase pension plan for her employees. She wants to fund the plan for each employee with either a term or whole life insurance policy. Which person does not pass the...
-
A retailing business imported food products for K50, 000 (CIF). Duty payable was 35%. The firm was registered for GST purposes. A. Calculate the final landed cost of the products, including GST. B....
-
2. The following data represent samples that were taken on 10 separate days. Each day has a varying sample size and the number of defects for the items sampled is listed. We want to see if this...

Study smarter with the SolutionInn App


