Use formal charge to choose the best Lewis structure for CH 3 SOCH 3 . H :O:
Question:
Use formal charge to choose the best Lewis structure for CH3SOCH3.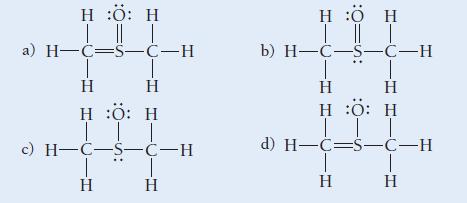
Transcribed Image Text:
H :O: H | || | a) H=C=5-C-H T Η Η H :O: H III © H-C-5-C-H Η Η H :O H | || | b) H-C-5-C-H Η Η H :O: H ||| d) H-C=s-C-H Η T Η
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
H O...View the full answer

Answered By

Mishark muli
Having any assignments and any other research related work? worry less for I am ready to help you with any task. I am quality oriented and dedicated always to produce good and presentable work for the client once he/she entrusts me with their work. i guarantee also non plagiarized work and well researched work to give you straight As in all your units.Feel free to consult me for any help and you will never regret
4.70+
11+ Reviews
37+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The algorithm below determines q, r EN such that y = q2 +r and r < 2, where y, z N. DIVIDE(y, z) 1 ry 2 q 0 3 4 5 6 7 8 9 10 11 12 while wy do w- 2w while w > z do q2q [w/2] W- if w
-
Briefly describe some common information system controls that need to be implemented by business managers, not IS professionals.
-
You have been assigned to a project to determine if a new investment should be made. your company uses a capital structure of 30% debt and 70% equity. the debt currently pays 8.5% interest; the...
-
Table illustrates the quantities, marginal costs, average variable costs, and average costs of a competitive firm. Refer to table 4. How much is the unit profit at price =$30? [the problem is based...
-
Describe some generic types of record keys in typical accounting databases. Are such keys simple or complicated?
-
What is the elasticity of supply of land within the borough limits of Manhattan?
-
Differentiate between trend analysis and comparative ratio analysis. Why is it useful to do a comparative ratio analysis? AppendixLO1
-
A depositor drew a check and delivered it to the payee. Fourteen months later, the check was presented to the drawee bank for payment. The bank had no knowledge that anything was wrong and paid the...
-
Return on Investment and Economic Value Added Calculations with varying Assumptions Knitpix Products is a division of Parker Textiles Inc. During the coming year, it expects to earn income of...
-
After her employer transferred her to another town, Rosemary put her house up for sale. After two years of Internet listings, open houses, repairs and price cuts, a buyer finally came along. By this...
-
Write a Lewis structure for the NO 3 ion. Include resonance structures.
-
The salinity of seawater can vary in the worlds oceans as shown in the map, which indicates salinity in units of percent by mass NaCl. Examine the image and answer the questions that follow. a. Which...
-
In problem, find the midpoint of the line segment joining the points P 1 and P 2 . P 1 = (-2, 0); P 2 = (2, 4)
-
Implement a Bayesian belief network in the programming language of your choice to represent a subject in which you are interested (for example, you might use it to diagnose medical conditions from...
-
Researchers have measured the acceleration of racing greyhounds as a function of their speed; a simplified version of their results is shown in Figure P4.67. The acceleration at low speeds is...
-
If the rate at which energy is dissipated by resistor 1 in Figure P31.86 is \(2.5 \mathrm{~W}\), and \(R_{1}=10 \Omega, \mathscr{E}_{1}=12 \mathrm{~V}\), and \(\mathscr{E}_{2}=6 \mathrm{~V},\) (a)...
-
Conservation efforts include reintroduction of species into the wild from captive breeding programs. Leung et al. (2018) rewilded mice from the inbred laboratory strain of mouse, C57BL/6, that had...
-
The ending balance of the Accounts Receivable account was \(\$ 7,800\). Services billed to customers for the period were \(\$ 21,500\), and collections on account from customers were \(\$ 23,600\)....
-
A gas mixture is found to contain two diatomic A and B species for which the partial pressures of both are 0.05065 MPa (0.5 atm). This mixture is to be enriched in the partial pressure of the A...
-
The following cost information was provided to you for analysis: September 12,000 Units Produced Costs: TIC TAC TOE TING August 10,000 P80,000 70.000 60.000 50,000 How much is the fixed cost per...
-
Consider the plane electromagnetic wave in vacuum (in SI units) given by the expressions E x = 0, E y = 2 cos [2 10 14 (t - x/c) + /2], and E z = 0. (a) What are the frequency, wavelength, direction...
-
Write an expression for the vector E- and vector B-fields that constitute a plane harmonic wave traveling in the +z-direction. The wave is linearly polarized with its plane of vibration at 45 to the...
-
Considering Eq. (3.30), show that the expression is correct as it applies to a plane wave for which the direction of the electric field is constant. KxE= (3.30) Ey 3 .
-
A semi-annual coupon bond has 15 years left to maturity. Its coupon rate is 6.5%. If you require an annual rate of return at 7% for your investment. What is this bond's intrinsic value to you? Please...
-
Steady Company's stock has a beta of 0.30. If the risk-free rate is 3% and the market risk premium is 8%, what is an estimate of Steady Company's cost of equity?
-
Meagan Dubean manages a portfolio of 200 common stocks. Her staff classified the portfolio stocks by 'industry sector' and 'investment objective.' Investment Industry Sector Objective Electronics...

Study smarter with the SolutionInn App


