Give the name or structural formula for each of the following ligands. Name acetylacetonate (a) (b) nitrite
Question:
Give the name or structural formula for each of the following ligands.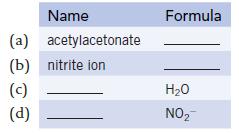
Transcribed Image Text:
Name acetylacetonate (a) (b) nitrite ion (c) (d) Formula H₂O NO₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Name Formula Ac...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Give the name or structural formula for each of the following ligands. Name ethylenediamine (a) (b) oxalate ion (c) (d) Formula NH3 SCN-
-
Define the following terms Chemistry, Organic Chemistry, Ionic and Covalent bond, Isomer, Hydrocarbons, Functional group, Clinical Chemistry, Inductive effect How Friedrich Whler synthesize the first...
-
Give the molecular formula for each of the following structural formulas. a. b. c. d. H-N-N-H hydrazine H-o-o-H hydrogen peroxide H-C-C-C-H H O H isopropyl alcohol Cl CI- P CI phosphorus trichloride
-
The following information pertains to the inventory of Parvin Company: Jan. 1 Beginning inventory 500 units 2,800 units Purchased Purchased 1,100 units Apr. 1 Oct. 1 $19 $24 $25 During the year,...
-
The materials used by the Toms River Division of Jadelis Company are currently purchased from outside suppliers at $35 per unit. These same materials are produced by Jadeliss Racine Division. The...
-
Most supermarkets across the United States have invested heavily in optical scanner systems to expedite customer checkout, increase checkout productivity, and improve product accountability. These...
-
Use the working papers and data from On Your Own 15-2. Forms for completing this On Your Own are provided in the Working Papers. Work independently to complete this problem. 1. Assume Cheries Pizza...
-
Rebeccas Pottery Loft makes a variety of handmade pottery items. She has asked for your advice on one of the items manufactureda clay pelican. The following information is provided: Number of...
-
A Firm has a stock price of 6 5 and an annual standard deviation of 2 5 % . The risk - free rate is 1 0 % ( c . c . ) . This firm will pay a dividend of $ 8 in exactly 3 months and no other dividends...
-
Give the oxidation number of the metal ion in each of the following compounds. (a) [Mn(NH 3 ) 6 ]SO 4 (b) K 3 [Co(CN) 6 ] (c) [Co(NH 3 ) 4 Cl 2 ]Cl (d) Cr(en) 2 Cl 2
-
One of the following nitrogen compounds or ions is not capable of serving as a ligand: NH 4 + , NH 3 , NH 2 . Identify this species, and explain your answer.
-
Jarvis Company wants to know if its profitability has improved. Calculate its profit margin for 2010 and 2009 using the following data: Net Income, 2010...........$ 10,000 Net Income,...
-
1. How much of the costs were allocated between the standard and deluxe models of the product? Is the marketing manager correct that LGI is making significant margins on the Deluxe Loot Box? Please...
-
Silver Titan's company has projected sales for the first seven months of 2021 are given below: Month January February March April May June July Amount $250,000 $340,000 $280,000 $300,000 $350,000...
-
Marcus purchased a diamond ring for $15,000 10 years ago. It was stolen in March this year. The ring was purchased to celebrate achieving a significant promotion at work. The FMV at the time of the...
-
Stan Rockford has the following passive activity items: Current-year passive income and loss amounts: $10,000 CHI ($7,000) JKL ($8,000) RST. What is the net income reported this year for the private...
-
On June 1st, Stiedemann entered into a contract to build a barn for Schaden : Base fee is $13,000 15% bonus for finishing 2 weeks early 5% bonus for finishing a week early Probability of finishing 2...
-
Using the present value of UTX's free cash flows calculated in problem 34 as a starting point, calculate the per share intrinsic value of UTX for each year 2010-2012.
-
Could the owner of a business prepare a statement of financial position on 9 December or 23 June or today?
-
A golf club head is made from aluminum having a specific weight of 0.100 lb/in 3 . In air it weighs 0.500 lb. What would be its apparent weight when suspended in cool water?
-
Wetsuits are prohibited in some triathlons due to the added buoyancy they provide the swimmer, essentially holding a greater portion of the body above the water and decreasing the power required to...
-
A cylinder that is 500 mm in diameter and 2.0 m long has a specific weight of 535 N/m 3 . It is held down into position with a cable attached to the sea floor. At this location, the sea is 500 m deep...
-
Use the following information about an interest rate SWAP contract to answer the following question. Assume for the date count fraction. (Do not round intermediate calculations.) If Bank of America...
-
Question 3 The number securities outstanding selling price The require rate of return Bonds 1,974 $ 1,096 10.08% Preferred stock 5,595 $85.58 19.45% Common Stocks 1,637 $115.52 13.22% Garden Tools...
-
A company manufactures and sells a single product. The variable cost of the product is $2.50 per unit and all production each month is sold at a price of $3.70 per unit. A potential new customer has...

Study smarter with the SolutionInn App


