Hydrazine, N 2 H 4 , can interact with water in two steps. (a) What is the
Question:
Hydrazine, N2H4, can interact with water in two steps.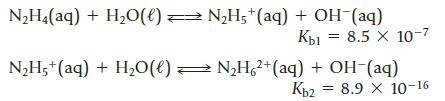
(a) What is the concentration of OH−, N2H5+, and N2H62+ in a 0.010 M aqueous solution of hydrazine?
(b) What is the pH of the 0.010 M solution of hydrazine?
Transcribed Image Text:
N2H4(aq) + H2O(0) = N2H5+(aq) + OH (aq) Кы 8.5 х 10-7 N2H5+(aq) + H2O(l) — N2H 2+(aq) + OH-(aq) Ког = 8.9 X 10-16
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To solve this problem well first calculate the concentrations of OH N2H5 and N2H62 in a 0010 M aqueo...View the full answer

Answered By

SUMAN DINDA
I LIKE TO TEACH STUDENTS. SO, I START MYSELF AS A PRIVATE TUTOR. I TEACH STUDENTS OF DIFFERENT CLASSES. I HAVE ALSO DONE BACHELOR OF EDUCATION DEGREE(B.ED). DURING THIS COURSE I HAD TO TEACH IN A SCHOOL. SO I HAVE A GOOD EXPERIENCE IN TEACHING.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Ethylenediamine, H 2 NCH 2 CH 2 NH 2 , can interact with water in two steps, forming OH in each step (Appendix I). If you have a 0.15 M aqueous solution of the amine, calculate the concentrations of...
-
Two 0.10-mol samples of the hypothetical monoprotic acids HA(aq) and HB(aq) are used to prepare 1.0-L stock solutions of each acid. a. Write the chemical reactions for these acids in water. What are...
-
Information is one of the most important assets of any business. Businesses must ensure information accuracy, completeness, consistency, timeliness, and uniqueness. In addition, business must have a...
-
Two independent situations follow: 1. Ready Car Rental leased a car to Culver Company for three months. Terms of the lease agreement call for monthly payments of $885, beginning on May 21, 2024....
-
Explain the difference between a firms corporate strategy and business strategy. Why do firms need to look at both aspects?
-
Adley Abdulwahab (Wahab) opened an account on behalf of W Financial Group, LLC, with Wells Fargo Bank. Wahab was one of three authorized signers on the account. Five months later, Wahab withdrew $...
-
Which data entry application control would detect and prevent entry of alpha betic characters as the price of an item in the inventory master file? a. field check c. completeness check b. limit check...
-
Calculating WACC Maxwell Industries has a debt-equity ratio of 1.5. Its WACC is 11 percent, and its cost of debt is 8 percent. The corporate tax rate is 35 percent. a. What is Maxwells cost of equity...
-
Establish your research (alternative) hypothesis and your null hypothesis for " If national norms indicated that college students tend to score about 4.0 on the college-specific anxiety scale...
-
Suppose a tank initially contains H 2 S at a pressure of 10.00 atm and a temperature of 800 K. When the reaction has come to equilibrium, the partial pressure of S 2 vapor is 0.020 atm. Calculate K p...
-
Ascorbic acid (vitamin C, C 6 H 8 O 6 ) is a diprotic acid (K al = 6.8 10 5 and K a2 = 2.7 10 12 ). What is the pH of a solution that contains 5.0 mg of acid per milliliter of solution? HO H H HO H...
-
Which are the most likely units for a measured temperature with a numerical value of \((a)-322\) and (b) 0 ?
-
Based on the topic below please provide the introduction and discussion in detail . Digital Marketing Before and after the Pandemic Introduction. Discussion Also provide a relevant topic related to...
-
A person exerts a 540 N force on the 80 kg block at an angle of 35 above the horizontal. The coefficient of friction is u = (0.3, 0.6). Make a complete model.
-
Using the topic of celebrities and influencers, provide a reflection of your understanding of the various marketing concepts as discussed in the unit BUS124 Global Marketing. Where applicable, your...
-
Hulu - Streaming Service. Hulu is not available in South Africa. A marketing plan for introducing Hulu to South Africa is an exciting exploration. The following is needed for a marketing plan to...
-
LA Gentry Can Company's (GCC) latest annual dividend of $1.40 a share was paid yesterday and maintained its historic 7 percent annual rate of growth. You plan to purchase the stock today because you...
-
A July 7, 2011 Pew Research Center poll asked a random sample of Americans to name the current news story that they were following the most at that time. The following table summarizes their...
-
After Theorem 1.5 we note that multiplying a row by 0 is not allowed because that could change a solution set. Give an example of a system with solution set S0 where after multiplying a row by 0 the...
-
a. The viscosity of Cl 2 at 293 K and 1atm is 132 P. Determine the collisional cross section of this molecule based on the viscosity. b. Given your answer in part (a), estimate the thermal...
-
a. The viscosity of O 2 at 293 K and 1atm is 204 P. What is the expected flow rate through a tube having a radius of 2.00 mm, length of 10.0 cm, input pressure of 765 Torr, output pressure of 760....
-
The Reynolds number (Re) is defined as Re = p(v x )d/, where and are the fluid density and viscosity, respectively; d is the diameter of the tube in which the fluid is flowing; and (v x )is the...
-
Kofi & Kofi Ltd uses the same amount of cash on each of the 250 days it operates during a year. Its annual demand for cash is estimated to be GH300,000 for the next financial year. Management...
-
Intro Tanrun Inc. is expected to pay an annual dividend of $0.45 per share in one year. Analysts expect the firm's dividends to grow by 6% forever. Its stock price is $37 and its beta is 1.7. The...
-
Your company reported the following information: Sales => $97,000,000 Cost of Goods Sold = 65% of Sales Operating Expenses Interest expense = = 5% of Sales $123,000 Operating capital = $10 million,...

Study smarter with the SolutionInn App


