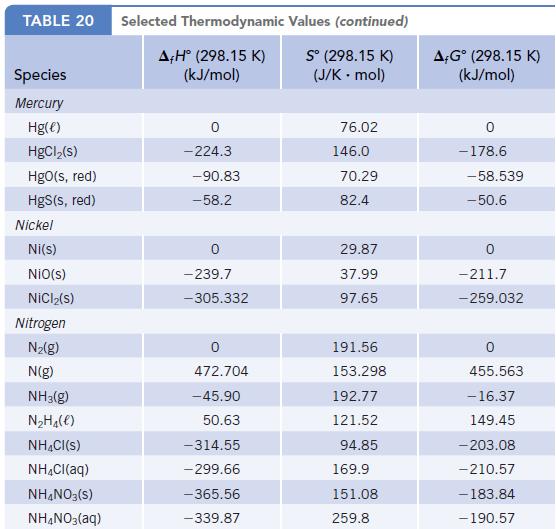
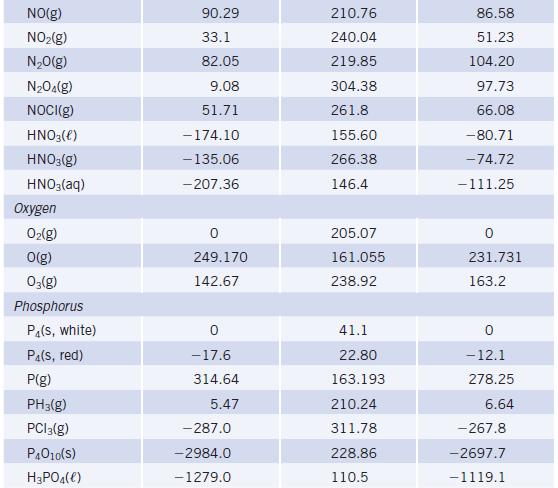
Use f H data in Appendix L to calculate the enthalpy change of the reaction Speculate
Question:
Use ΔfH° data in Appendix L to calculate the enthalpy change of the reaction![]()
Speculate on whether such a reaction could be used to “fix” nitrogen. Would research to find ways to accomplish this reaction be a useful endeavor?
Data given in Appendix L
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:





