You are asked to identify an unknown liquid that is known to be one of the liquids
Question:
You are asked to identify an unknown liquid that is known to be one of the liquids listed below. You pipet a 3.50-mL sample into a beaker. The empty beaker had a mass of 12.20 g, and the beaker plus the liquid weighed 16.08 g. 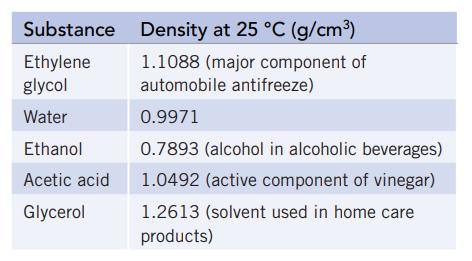
(a) Calculate the density and identify the unknown.
(b) If you were able to measure the volume to only two significant figures (that is, 3.5 mL, not 3.50 mL), will the results be sufficiently accurate to identify the unknown? Explain.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:





