The gas-phase reaction of nitrogen dioxide with fluorine proceeds to form nitrosyl fluoride, NO 2 F. Determine
Question:
The gas-phase reaction of nitrogen dioxide with fluorine proceeds to form nitrosyl fluoride, NO2F.![]()
Determine whether the following mechanism provides the correct overall stoichiometry, and identify intermediates, if any.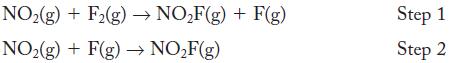
Strategy
Sum the proposed elementary steps to determine whether these steps produce the overall stoichiometry.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:





