A gas stream of flow rate 10.0 lbmole/ft 2 · h contains 6.0% sulfur dioxide (SO 2
Question:
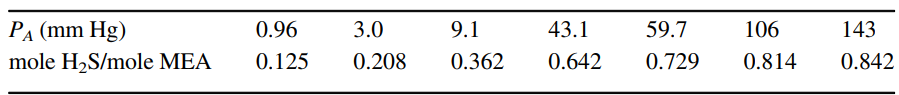
a. Specify the molar flow rate and mole fraction composition of all terminal process streams for operation at 2.0 times the minimum solvent flow rate.
b. Determine the height of packing required to accomplish the separation or operation at 2.0 times the minimum solvent flow rate.
Equilibrium distribution data for SO2 in water at 30°C (A = SO2):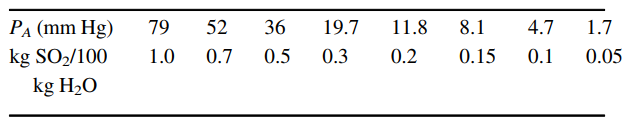
The word "distribution" has several meanings in the financial world, most of them pertaining to the payment of assets from a fund, account, or individual security to an investor or beneficiary. Retirement account distributions are among the most...
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Fundamentals Of Momentum Heat And Mass Transfer
ISBN: 9781118947463
6th Edition
Authors: James Welty, Gregory L. Rorrer, David G. Foster
Question Posted:





