Four vapor pressure data pointstwo representing solidvapor equilibrium and two representing liquidvapor equilibriumare available for a compound:
Question:
Four vapor pressure data points—two representing solid–vapor equilibrium and two representing liquid–vapor equilibrium—are available for a compound:
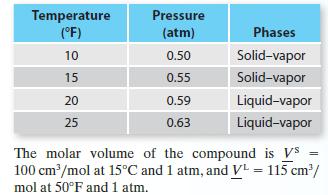
A. Give your best estimate of the triple point pressure and temperature for this compound.
B. Give your best estimate of pressure at which solid–liquid equilibrium occurs at T = 25°F.
C. Give your best estimate of the normal boiling point.
Transcribed Image Text:
Temperature (°F) 10 15 20 25 Pressure (atm) 0.50 0.55 0.59 0.63 Phases Solid-vapor Solid-vapor Liquid-vapor Liquid-vapor The molar volume of the compound is Vs = 100 cm³/mol at 15°C and 1 atm, and V¹ = 115 cm³/ mol at 50°F and 1 atm.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
Four vapor pressure data pointstwo representing solidvapor equilibrium and two representing liquidvapor equilibriumare available for a compound: A. Give your best estimate of the triple point...
-
THIRD AVENUE SOFTWARE HEALTH-CARE APP PROJECT This case is new for the ninth edition of Information Technology Project Management . The case provides an opportunity to apply agile and Scrum...
-
A compound has a molecular mass of 120 g/mol, and the information in the table below is the only other data available for a compound. Fill in all of the empty cells with your best estimate of the...
-
Methanol, CH3OH, is prepared industrially from the gasphase catalytic balanced reaction that has been depicted here using molecular models. In a laboratory test, a reaction vessel was filled with...
-
1. Compare GEs approach to performance evaluation with that of ICI (mentioned in the chapter). 2. Critically evaluate the strengths and weaknesses of each companys approach to the performance...
-
Determine the upper tail critical value of F in each of the following one tail tests. a. = 0.05, n1 = 16, n2 = 21 b. = 0.01, n1 = 16, n2 = 21
-
18.7 What is the direct materials price variance for the month? a) 2 226 (F) b) 2 226 (A) c) 2 385 (F) d) 2 385 (A).
-
The following selected data were taken from the financial statements of The OMalley Group Inc. for December 31, 20Y5, 20Y4, and 20Y3: No dividends on common stock were declared between 20Y3 and 20Y5....
-
Troy Company earned $15,000 of cash revenue. Troy incurred $12,000 of utility expense on account during Year 1. The company made cash payments of $8,000 to reduce its accounts payable during Year 1....
-
This problem involves the same compound that was examined in Problems 6-14 through 6-17, which in the vapor phase was described by the EOS: A. The fugacity in the vapor phase at T = 50C and P = 0.1...
-
Estimate the vapor pressure of ethanol at temperatures of T = 0, 50, 100, and 150C, using the following methods. A. The Antoine equation B. The Clausius-Clapeyron equation with H vap = 42.0 kJ/mol...
-
Use a calculator to approximate each logarithm to four decimal places. log 90
-
1. Allen Young has always been proud of his personal investment strategies and has done very well over the past several years. He invests primarily in the stock market. Over the past several months,...
-
The waiting times between a subway departure schedule and the arrival of a passenger are uniformly distributed between 0 and 9 minutes. Find the probability that a randomly selected passenger has a...
-
Greenview Dairies produces a line of organic yogurts for sale at supermarkets and specialty markets in the Southeast. Economic conditions and changing tastes have resulted in slowing demand growth....
-
Rudy Gandolfi owns and operates Rudy's Furniture Emporium Inc. The balance sheet totals for assets, liabilities, and stockholders' equity at August 1, 2019, are as indicated. Described here are...
-
If you were team leader how would you break up this assignment for 4 people to complete? Group Case Analysis Parts 4, 5, and 6 IV. STRATEGY IMPLEMENTATION. (How are you going to do what you want to...
-
A plastic beach ball has radius 20.0 cm and mass 0.10 kg, not including the air inside. (a) What is the weight of the beach ball including the air inside? Assume the air density is 1.3 kg/m 3 both...
-
An auto-parts manufacturer is considering establishing an engineering computing center. This center will be equipped with three engineering workstations each of which would cost $25,000 and have a...
-
The following represents pressure samples, in pounds per square inch (psi), taken in a fuel line once every second for 10 sec. a. Fit a first-degree polynomial, a second-degree polynomial, and a...
-
A liquid boils when its vapor pressure equals the external pressure acting on the surface of the liquid. This is why water boils at a lower temperature at higher altitudes. This information is...
-
The solubility of salt in water is a function of the water temperature. Let S represent the solubility of NaCl (sodium chloride) as grams of salt in 100 g of water. Let T be temperature in C. Use the...
-
You are the audit team assign to audit the unaudited set of financial statements for XYZ Company Ltd. Prepare written report to highlight the audit procedures to be used to substantiate/confirm the...
-
Island Novelties, Inc., of Palau makes two products--Hawaiian Fantasy and Tahitian Joy. Each product's selling price, variable expense per unit and annual sales volume are as follows: Selling price...
-
Fantastic Fashions has just completed its first quarter of operations. Assume that Fantastic Fashions adjusts its book quarterly. Below are transactions that have not yet been recorded. Jan 1 Made...

Study smarter with the SolutionInn App


